
Abstract
Polymorphism of the catalase gene, and the activity of erythrocyte SOD and catalase. The subjects were stable COPD patient ever smokers (n5165) and healthy controls, matched for age and cigarette consumption. Genotyping of Mn-SOD at Ala16Val and the catalase gene at-262C.T was performed, and the functional activity of SOD and catalase in.
The pathogenesis of Alzheimer’s disease (AD) remains to be elucidated. Oxidative damage and excessive beta-amyloid oligomers are components of disease progression but it is unclear how these factors are temporally related. At post mortem, the superior temporal gyrus (STG) of AD cases contains plaques, but displays few tangles and only moderate neuronal loss. The STG at post mortem may represent a brain region that is in the early stages of AD or alternately a region resistant to AD pathogenesis. We evaluated expression profiles and activity of endogenous anti-oxidants, oxidative damage and caspase activity in the STG of apolipoprotein ε4-matched human AD cases and controls. Total superoxide dismutase (SOD) activity was increased, whereas total glutathione peroxidase (GPX), catalase (CAT) and peroxiredoxin (Prx) activities, were decreased in the AD-STG, suggesting that hydrogen peroxide accumulates in this brain region. Transcripts of the transcription factor NFE2L2 and inducible HMOX1, were also increased in the AD-STG, and this corresponded to increased Nuclear factor erythroid 2-related factor (NRF-2) and total heme-oxygenase (HO) activity. The protein oxidation marker 4-hydroxynonenal (4-HNE), remained unchanged in the AD-STG. Similarly, caspase activity was unaltered, suggesting that subtle redox imbalances in early to moderate stages of AD do not impact STG viability.
Introduction
Alzheimer’s disease (AD) is the most common neurodegenerative disease. Its prevalence is increasing markedly worldwide due to ageing populations and a lack of disease modifying therapies. AD clinically manifests as memory deficits in combination with a progressive deterioration of other cognitive domains such as executive function and visuospatial skills.
At post mortem examination severely affected AD brains are commonly reduced to half their original volume1 with a disproportionate neuronal loss from the entorhinal cortex, amygdala, and hippocampus2. The pathology of AD is also characterized by two pathognomonic entities, intracellular neurofibrillary tangles (NFTs) and extracellular plaques. The major constituent of plaques is a collection of peptides called beta-amyloid (Aβ) while NFTs are largely composed of paired helical filaments of an abnormally hyperphosphorylated form of the microtubule associated protein tau (MAPT or tau). Tau pathology spreads in a predictable manner, from the allocortex to the association areas of the neocortex and lastly the primary cortices3,4. Cortical layers within the entorhinal cortex from severely affected subjects can exhibit up to 90% neuronal loss5 while regions such as the primary visual cortex remain essentially unaffected by the disease process with mild Aβ accumulation and minor alterations in neuronal form and functions.
The popular amyloid cascade hypothesis (ACH), suggests that the common sporadic forms of AD results from the accumulation of soluble oligomeric forms of Aβ in the neuropil. These toxic products disrupt neuronal kinase/phosphatase and redox balance, leading to tau hyperphosphorylation, NFT formation and neurodegeneration6. However, the mechanisms leading to the accumulation of Aβ oligomers in sporadic AD are not clear. Epidemiological studies have not been particularly informative with only ageing, the possession of the apolipoprotein E (APOE) ε4 allele, female gender and diabetes being consistently identified as risk factors7.
In addition to plaques and NFTs, the AD brain is also characterized by oxidative damage to proteins, nucleic acids8 and lipids9. It is generally accepted that Aβ may act directly as a pro-oxidant10 or indirectly by precipitating NMDA receptor-dependent Ca2+ influxes that result in mitochondrial dysfunction and the subsequent generation of reactive oxygen species (ROS)11. These, in turn, stimulate antioxidant response pathways that involve redox sensitive transcription factors e.g., NRF-2 (nuclear factor, erythroid 2-like 2; encoded by NFE2L2). Activation of NRF-2 is known to stimulate the potent inducible antioxidant hemeoxygenase-1 (HO-1) and possibly other endogenous antioxidant response elements11. Yet other researchers hypothesise that Aβ accumulation may be a consequence of oxidative stress12 and suggest that Aβ13 and indeed tau14 act as anti-oxidants in AD (reviewed by Sutherland et al.)15 thereby, potentially limiting the observed radical-mediated damage to DNA, proteins and lipids that occurs during the pathogenesis of this neurodegenerative disease16,17.
Notwithstanding that the outcomes from clinical trials of Aβ-modifying therapies in asymptomatic individuals are outstanding, there is still a need to pursue adjunctive or alternative therapeutic targets in AD, through accurate modelling of the early phases of the disease18. In this study, we explore markers of oxidative damage in the superior temporal gyrus (STG). The AD-STG at post-mortem shows moderate levels of insoluble tau and plaques while retaining the majority of its neurons19. Therefore, the post-mortem STG may represent a surrogate for the state of the most severely affected regions such as the entorhinal cortex, earlier in the disease course. An understanding of events in the post-mortem STG may allow a better temporal appreciation of linkages between oxidative stress and AD pathogenesis and more generally reveal novel pathways to exploit in the development of therapeutics to combat AD at a stage prior to irreversible cognitive decline.
Results
Detection of advanced Aβ but early tau pathology in AD-STG
As previously described, the STG region of the AD cases (AD-STG) in this cohort showed typical AD pathology with significantly more plaques (both diffuse and cored) and AT8-positive neurons (Fig. 1A,B and Supplementary Table 1)19. Similarly, immunoblotting showed markedly increased AT8 tau-positive bands (between 45–79 kDa) in the AD-STG (Fig. 1B, refer to Supplementary Fig. S1). NFTs were relatively sparse, and neuronal counts were lower (16%) but not significantly different to controls (Supplementary Table 1). Taken together, the paucity of NFTs and minimal loss of neurons suggest that the disease, tau pathology in particular, is at an early stage in the AD-STG compared with the medial temporal lobe structures (Braak stage V/VI) from these same patients. AD cases also showed a significantly lower mean RNA integrity number (RIN) and brain pH and these two parameters were correlated with one another (r2 = 0.2, p = 0.002) (Supplementary Table 1). Although no differences in the mean fixation (storage) time (p = 0.06) between AD cases and controls were noted, fixation time was noted to range from 6 to 155 months. Additionally, a difference in post-mortem interval (PMI) was observed between the two groups (Supplementary Table 1; p = 0.04).
Assessing anti-oxidant gene expression in AD and control STG
The disparity in mean RNA integrity number (RIN) identified between the cases and controls had the potential to skew gene expression analyses. In particular housekeeping genes need to reflect RIN differences between cases and controls rather than no difference as per normal19. The housekeeping gene Succinate Dehydrogenase Complex Flavoprotein Subunit A (SDHA) satisfied this modified criterion while displaying no difference between cases and controls in a RIN-matched subcohort (n = 21, data not shown). With SDHA as house-keeping gene, significant increases in both NFE2L2 (encoding the transcription factor NRF-2) (p = 0.008) and HMOX1 (encoding HO-1, the inducible form of HO) (p = 0.009) mRNA were determined in the AD-STG (Table 1). In contrast, there were no differences in HMOX2 (encoding the constitutively expressed form of HO, HO-2) or SOD1 mRNA with the latter varying widely in the AD-STG.
Anti-oxidant protein levels and activity
There was no difference in SOD1, SOD2 and GPX1 protein levels between AD and control groups as judged by direct ELISA (Fig. 2A). However, total SOD activity increased significantly (p = 0.04) (Fig. 2B), while total GPX activity was decreased in the AD-STG compared to controls (p = 0.02) (Fig. 2C). This data was consistent with an imbalance in H2O2 producing vs consuming enzymatic activity and warranted further investigation to assess other endogenous H2O2-consuming enzymes.
Surprisingly, the capacity for the AD-STG to degrade exogenously added peroxides was slightly greater than control-STG (p = 0.002), as judged by an ex vivo assays measuring rates of H2O2 consumption (Fig. 3A). To understand the specific contribution of CAT to this peroxide consuming activity in control and AD brains, H2O2 decomposition by added STG-homogenate was reassessed in the presence of aminotriazole (AT), a CAT-inhibitor. Under these conditions, H2O2 consumption diminished more in the control group than in the AD cohort, suggesting a higher level of catalase specific activity in control STG (i.e., compared the difference in activity between the white bars (control) and then the black bars (AD) in the absence and presence of AT, Fig. 3A). Overall, we determined a 1.5-fold lower CAT activity in the AD-STG than the corresponding enzymatic activity in the control-STG (p < 0.001) (Fig. 3B). This decreased CAT activity exacerbates the documented loss of GPX activity in the same STG tissues (Fig. 2C) and together imply that the AD-STG shows a limited ability to enzymatically decompose accumulating H2O2.
Overall, total consumption of H2O2 is likely to be mostly ascribed to a combination of CAT and heme-peroxidase activity in the clarified brain homogenates as sodium azide (SA; a highly active heme-peroxidase inhibitor)20, completely inhibited peroxide consumption in both the control and AD cohorts (Fig. 3A). Note, although azide is a potent inhibitor of catalase activity, azide-insensitive catalase activity has been reported in bacteria21, and this may impact on the interpretation of these inhibition studies.
The peroxiredoxin (Prx) family of anti-oxidant enzymes, are also a pathway for eliminating H2O2, and have also been implicated in the pathogenesis of AD22. Therefore, we measured total peroxiredoxin activity by assessing the selective consumption of cumene-hydroperoxide. Similar to catalase and total GPX activity, peroxiredoxin activity decreased slightly (and significantly) (p = 0.004) in the AD-STG compared to the controls (Fig. 3C). Together these data indicate that the AD-STG is less capable of detoxifying accumulating (low molecular weight) peroxides via multiple antioxidant enzymic pathways compared with the corresponding control tissue.
Additionally, total and cytosolic NRF-2 protein expression was assessed using a combination of immunohistochemistry and western blotting respectively. Consistent with the gene response, a statistically significant increase in total NFE2L2 expression was observed in the AD cohort (p = 0.05) (Fig. 4A–C). To assess potential differences in NRF-2 subcellular localisation, NRF-2 protein levels were also assessed in cytosolic homogenate fractions. A recent consensus in the literature indicates that the molecular weight for NRF-2 occurs between 95–100 kDa23 therefore, bands detected at mass 95 kDa were used quantify a significant decrease in cytosolic NRF-2 levels in the AD cohort (p = 0.004) (Fig. 4D and E, refer to Supplementary Fig. S2), suggesting that NRF-2 levels are inherently lower in the AD-STG and/or that NRF-2 may be sequestered to the nucleus in this cohort.
Total HO activity, a downstream transcriptional target of NRF-2, was also assessed in the homogenized brain tissues. Overall, we determined a significant increase in total HO activity, as indicated by increased bilirubin production in the ex vivo assay (p = 0.04) (Fig. 5; see Supplementary Fig. S3 for exemplar chromatograms). This result taken together with the corresponding gene studies that showed a selective increase in the inducible HMOX1 mRNA but not HMOX2 mRNA, suggest that active HO-1 protein is expressed in the AD-STG and this likely accounts for the parallel increase in total HO activity in these same tissues.
Assessing oxidative damage in the AD-STG
Oxidative tissue damage was determined by immunostaining for 4-hydroxynonenal (4-HNE)+. There were no difference between cases and controls (Fig. 6A,B) suggesting that despite the documented decrease in antioxidant enzymes responsible for detoxifying H2O2, oxidative protein damage remained unchanged. Semi-quantitation of the 4-HNE+ staining confirmed no difference in levels of this biomarker of oxidation (Fig. 6C). While this specific form of oxidized damage was unaffected, the altered patterns of anti-oxidant response elements documented in the AD-STG are consistent with these tissues experiencing a subtle heightened oxidative stress.
Caspase-dependent apoptosis
To investigate whether changes in H2O2 in the AD-STG promoted pro-apoptotic cell death pathways, caspase 3/7 activity was determined in control- and AD-STG homogenates. This biomarker of apoptosis was marginally higher in the AD-STG homogenates relative to the controls, although this did not reach statistical significance (p = 0.08) (Fig. 7). Therefore, our data are consistent with unaltered capsase-mediated cell death (i.e., intrinsic apoptosis) in the AD-STG.
Correlations between biochemical markers and AD pathology
Overall there was a paucity of correlations identified and some inconsistency in the relationships using different biomarkers of AD. From those that showed linear relationships total HO activity was significantly and positively correlated with STG plaque areal fraction (p = 0.04, r2 = 0.21) (Fig. 8A) as well as total NFTs in the AD-STG (r2 = 0.24, p = 0.03) (Fig. 8B). In addition, total SOD activity was positively correlated with total NFTs (r2 = 0.23, p = 0.03) (Fig. 8C), while total CAT (Fig. 8D) and Prx (p = 0.02, r2 = 0.26) (Fig. 8E) activities were negatively correlated with AT8-tau positive cells.
Interestingly no relationships between the areal fraction of plaques and any other antioxidant protein activities were identified in the brain tissues analysed here. Nor were plaques related to cytosolic or total NRF-2 expression (see Supplemental Fig. S4). Also, we found no significant correlations between AT8-tau and total NFT immunoreactivity or either of these pathological tau indices and biochemical activity of the antioxidant enzymes (Supplemental Figs S5 and S6 respectively).
Discussion
Despite its inherent retrospective nature post-mortem brain tissue remains the primary vehicle for mechanistic investigations in humans. We have previously suggested that investigating the less affected areas of the AD brain may represent a surrogate for more severely affected areas at an earlier stage in the pathogenesis of AD24. Albeit it is also possible that the STG is resistant to AD rather than belatedly affected. In an effort to understand early molecular events associated AD we investigated the STG, a region that is moderately affected in the AD brain with characteristic Aβ pathology, including neuritic plaques, but only mild tau-related changes and minor neuronal loss.
The popular ACH states that a build-up of soluble Aβ oligomers in the brain parenchyma triggers a sequence of neurotoxic events, including oxidative stress that eventually leads to NFT formation and neuronal death25. An alternative hypothesis has been proposed that places Aβ accumulation secondary to oxidative stress26. Here we specifically hypothesized that examining oxidative stress in a belatedly affected area of the AD brain (as evidenced primarily by Aβ accumulation) would allow us to help clarify the temporal relationship of oxidative stress in AD pathogenesis. The majority of our findings appear to support an increase in oxidative stress with plaques being associated with subtle changes in redox balance insufficient to result oxidative lipid/protein damage.
Attenuation of the antioxidant actions of peroxide-consuming enzymes CAT, GPX and peroxiredoxins with a parallel increase in SOD activity (responsible for converting the superoxide radical anion to H2O2) likely creates an intracellular milieu containing excess peroxides in the AD-STG. Although we were able to show decreases in specific peroxidase and catalase activity, the total H2O2 consumption measured in STG clarified homegenates was initially higher in AD-STG. We employed a strategy using specific inhibitors or peroxide substrates to evaluate contributions of GPX, CAT and peroxiredxin activities, which were all determined to be lower in the AD-STG. Thus, enhanced dismutation of superoxide radical anion leads to elevated H2O2 that is ineffectively degraded by virtue of decreased capacity for the STG to consume this pro-oxidant. That the total H2O2 consumption was initially higher in the AD-STG than in control-STG requires further discussion. This apparent paradox may reflect methodological limitations or that H2O2 is consumed by reactions with unbound metal ions released through tissue processing. Others have demonstrated that transition metals increase around plaques and NFTs in the AD brain27. These transition metals may contribute to the consumption of H2O2 to yield other ROS species28 rather than being degraded to benign products. For example, Fe2+ and H2O2 yield the highly reactive ROS, OH• via the Fenton or Haber–Weiss reactions15. However, our data indicating that sodium azide treatment inhibited the majority of ex vivo H2O2 consumption, strongly suggesting that free metals play only a minor role in this process, with a majority of transition metals likely to remain bound to proteins and unable to participate in unregulated redox reactions. Furthermore, the increased consumption of exogenous peroxide is unlikely to reflect a methodological limitation as the outcome from the studies with the heme-poison sodium azide points to other pathways of ex vivo H2O2 consumption linked to other peroxidases that are present in brain tissues and not identified here.
A study by Gsell and colleagues provides further evidence for an accumulation of H2O2 in the AD brain, as judged by assessments of the ratio of SOD/CAT activity. This study reported that SOD activity remained unchanged in the AD brain, however similar to outcomes we document here, a decreased CAT activity yielded an overall increase in oxidative stress through an accumulation of H2O2. These results were consistent in multiple post-mortem brain regions, ranging from mild to severely affected regions of AD29 and we have built on this outcome by demonstrating decreased activity for other enzymes responsible for H2O2 consumption in the AD brain. Another study reports decreased mitochondrial SOD 2 levels and unchanged SOD1 levels in patient serum, relative to age and gender matched controls30. While any deviation from age and gender matched controls may still reflect altered redox homeostasis in AD, differences in SOD levels reported in our present study may reflect difference in redox states between the peripheral and central nervous system of early stage AD patients. Notably, small sample size, has been suggested to contribute to the inconsistent SOD serum levels that have been reported in the literature (reviewed by Chang et al.31). Thus, Chang and colleagues identified that the three studies with over 80 cases all report significantly increased SOD levels. Moreover, differences in SOD levels may reflect different disease stages. Consistent with our biochemical outcomes measured in the STG, which reflects the early changes in AD etiology, comparisons of leukocyte SOD levels in patients with severe and mild AD vs corresponding control subjects suggest that SOD levels are elevated in the early stages of AD, however appear to be depleted in the later stages of disease progression32. Although our data suggests a likely accumulation of H2O2 in the AD-STG, we found no evidence of increased ROS-mediated damage at least in the form of 4-HNE+ immuno-staining. As previous reports indicate that differences in tissue-storage time influence immuno-staining intensities33, and so may impact on the interpretation of this immune-staining approaches, 4-HNE+ immuno-staining was repeated with a sub-cohort that were more closely matched for storage time; no difference in result was obtained. Despite no change in 4-HNE+, we chose to assess additional oxidative markers to corroborate this outcome and demonstrate oxidative stress in the AD-STG.
The transcription factor NRF-2 is now considered a master redox switch that initiates a suite of cyto-protective genes that are central to cell survival34. Both gene and possibly nuclear localisation of NRF-2 were increased in the AD-STG, suggestive of stress-induced NRF-2 activation early in AD pathogenesis. By contrast, studies using highly affected areas of the AD brain suggest that nuclear levels, and thus activation, of NRF-2 decrease in the AD brain35,36. This suggests that NRF-2 protein levels may decrease with increasing severity of AD pathology. Overall, our data together with outcomes from others37,38 provide further evidence to suggest that therapies targeting an enhancement of NRF-2 dependent pathways may serve as a promising treatment for pre-symptomatic AD subjects.
Here we found an increase in expression of inducible HO-1 protein; a downstream target of transcriptional NRF-2. As levels of HMOX-2 mRNA remained unchanged the simplest explanation for the observed increase in total HO activity, measured in an ex vivo assay employing homogenates from AD-STG, is likely attributed solely to alteration in the inducible HO-1 protein. Although previous studies using AD hippocampal tissues also reported increased HO-1 expression, this has not translated to increased activity or neuroprotection39.
While our data suggest that total HO activity may be enhanced and protective early in the development of AD pathology, it must be recognized that our assessment of HO activity was determined using an ex vivo assay that added liver-derived biliverdin reductase to convert biliverdin into bilirubin, with the latter biomarker of HO activity detected by liquid chromatography. To add biological context to our result, recent studies have indicated decreases in biliverdin reductase activity with increasing severity of AD pathology40,41 thereby potentially limiting the conversion of biliverdin to the more potent antioxidant bilirubin. Limitations in the conversion of biliverdin to bilirubin may therefore be an important feature of whether HO-1 confers neuro-protection in different individuals, and in turn, potential resistance to developing advanced AD pathology.
Stocker and colleagues42,43 have demonstrated soluble bilirubin to be a potent inhibitor of lipid peroxidation. Also, inhibition of HO-1 activity in stress induced endothelial cells has demonstrated increased sensitivity to ROS mediated damage, with restored effects of cell viability attributed to the addition of bilirubin44. A gene deletion of biliverdin reductase has further been associated with increased endogenous oxidative stress45, suggestive of a direct role for bilirubin in conferring protection by HO-1, albeit that carbon monoxide, another by-product of the catabolic heme pathway can also confer neuroprotection46.
Our data suggests a requirement for a functional HO/BV/BR system and that augmenting this pathway in AD may be a target for future therapy. However, while the NRF-2/HO-1 pathway may seem an attractive therapeutic target for AD47 a comprehensive understanding of mechanisms regulating HO-1 expression in brain cells, whether other enzymes such as biliverdin reductase are active, and how these mechanisms link to neuropathological changes is essential before this pathway can be exploited for therapeutic development. Consistent with this conclusion, studies with a transgenic AD mouse model demonstrate48,49 increased levels of oxidative stress, AD pathology and enhanced cognitive dysfunction when double crossed with a knockout of antioxidant defences, including deficiencies in SOD50,51, and GPX52, while overexpression of SOD appears to reduce ROS levels, memory deficits and plaque load in respective AD mice48,49. Similarly, inducing NRF-2 activity in transgenic AD mice yields enhances neuroprotection against oxidative stress attributed to increased HO-1 expression53,54.
On balance the majority of data obtained here suggest that oxidative stress is subtly elevated in the AD-STG, and potentially in the early stages of AD pathogenesis, as judged by altered activity of a number of antioxidant response elements albeit in the absence of detectable oxidative protein damage. The limited extent of pathology and largely preserved complement of neurons observed in the STG suggests that a degree of resistance to oxidative stress is operative. Our findings are consistent with a mechanism whereby the STG, relative to more severely affected areas in the medial temporal lobe, retains the ability to dismutate the superoxide radical anion into H2O2, stimulating NRF-2 to increase total HO-1 and perhaps BVR activity to ultimately resist neuronal loss that is characteristic of AD.
Methods
Case selection
As previously described19 frozen and fixed specimens of superior temporal gyrus (STG) from 21 age, gender- and APOE ε4-matched AD cases and 19 neurologically normal controls were obtained from New South Wales Brain Banks (NSWBB) following approval by the University of Sydney’s Human Research Ethics Committee (#HREC 11245/USYD 12734) and the NSW Scientific Advisory Committee. Pathological AD was diagnosed by a neuropathologist using standard diagnostic criteria55 and cases were either Braak stage IV (n = 1), V (n = 4) or VI (n = 12); four cases did not fit staging criteria (see Supplementary Table S1). Brains were from subjects that were to the best of our knowledge unaffected by any infectious disease, or other known neurological disorders. Any brain sample obtained with post-mortem intervals (PMI) > 2.6 days or significant agonal events were excluded from this study. Control brain tissue employed here was derived from a cohort of coronial cases, with the primary mode of death recorded as myocardial infarction.
Tissue preparation
Samples of frozen brain tissue (30 mg) were suspended in 600 μL lysis buffer containing 6 μL beta-mercaptoethanol as provided by the Bioline Isolate II RNA Kit (Bioline, Australia; BIO-5207) then homogenized with a rotating piston arrangement (Wheaton Specialty Glass, NJ, USA) at 500 r.p.m.56, and centrifuged (5 min, 13,000 × g) prior to RNA extraction. Total RNA was extracted according to the manufacturer’s instructions then quantified using a Nanodrop ND-1000 spectrophotometer (Thermo Scientific, Wilmington, USA); RNA quality was determined using a BioAnalyzer 2100 (Agilent Technologies, Santa Clara, USA) and individual RIN values shown in Supplementary Table S1. All samples were stored at −80 °C prior to use.
Additional samples of frozen human brain STG tissue (~100 mg) were cut into small pieces, suspended in 2 mL Buffer (50 mM PBS, pH 7.4) containing: 1 mM EDTA, 10 μM butylated hydroxytoluene and a Protease Inhibitor Cocktail tablet (Roche Diagnostics, Bern Switzerland; 1 tablet/50 mL buffer). The suspension was homogenized as described above and the homogenates divided into 0.2 mL aliquots, immediately snap frozen in liquid nitrogen and stored at −80 °C. As required, homogenates were thawed at 20 °C then centrifuged (5 min, 13,000 × g) to clarify the supernatant for use in all biochemical and immune-assays (see below). Protein concentrations for all samples were determined using the Bicinchoninic Acid (BCA) assay (Sigma-Aldrich, St Louis, USA) and average values used to normalize all subsequent biochemical parameters measured.
Materials
Chemicals were obtained from Sigma-Aldrich (Castle Hill NSW 1765, Australia), unless stated otherwise. All solutions were freshly prepared using MilliQ® Water or high quality analytical grade organic solvents and, where appropriate, sterilized prior to use.
Gene expression
Complementary DNA (cDNA) was synthesized using the Tetro cDNA Synthesis Kit (Bioline, Australia; BIO-65043) and gene expression was determined using RT-qPCR performed with a Roche LightCycler 480 system (Roche Diagnostics Corporation, Indianapolis, Indiana, USA) according to MIQE recommendations57 as previously described19. Primer efficiencies and sequence pairs used for all RT-qPCR analyses are shown in Table 2. Melt curves were generated using the LightCycler 480SW v1.5.1 software to demonstrate a single PCR product (see Supplementary Fig. S7). RT-qPCR analysis was carried out using the GenEx qPCR data analysis software (v5.3.2) (MultiD Analyses, Goteborg, Sweden). Importantly, correlations were performed to assess the impact of RNA integrity number (RIN) and tissue pH on expression levels of the genes of interest studied here as described in detail elsewhere19. This approach afforded the selection of a suitable reference gene that normalized RIN/pH differences between case and control tissue samples.
Immunohistochemistry
Briefly, formalin-fixed, paraffin embedded (FFPE) sections were obtained from the contralateral STG of all individuals. FFPE-STG sections (thickness 10 μm) were stained with cresyl violet and Garvey-modified silver stain or immunostained for phospho-tau (AT8, mouse monoclonal, Thermo Scientific, MI161393)19.
In addition, FFPE-STG sections were immunostained for 4-hydroxy-2-nonenal (4-HNE+) and NRF-2. Briefly, sections were rehydrated and antigen retrieved using a decloaking chamber (Biocare Medical, CA, USA). Samples were heat retrieved at 95 °C for 30 min in a pH 9 buffer (Agilent Technologies, Santa Clara, CA, USA, #S2367), cooled to 23 °C and treated with 3% v/v H2O2 in dH2O to block for endogenous peroxidase activity. Sections were then incubated with a serum-free protein block (Agilent Technologies, #X0909) for 30 min, followed by application of primary antibody including: a polyclonal anti-4-HNE antibody (1:200 v/v; overnight incubation at 4 °C) (Bioss, Boston, USA, #bs-6313R) and an oligoclonal anti-NRF-2 antibody (1:200 v/v, 1 h incubation at 23 °C) (Thermo Fisher Scientific, MA, USA, #710574). Products were then visualized via incubation with the DAKO EnVision™ + System-HRP (Agilent Technologies, #K4002; 30 min at 23 °C), followed by a 5 min incubation with DAB (Agilent Technologies, #K3468). Sections were counterstained using hematoxylin, dehydrated and mounted with DPx.
The oxidation biomarker 4-HNE was selected as a measure for tissue oxidation status as critically reviewed by Esterbauer and colleagues58. The 4-HNE immuno-sections were imaged simultaneously using the same parameters with an Axio Scan.Z1 slide-scanning microscope (Carl Zeiss, Jena, Germany). Entire sections were scanned using a 10x objective and imported into MetaMorph software program (v7.6 Universal Imaging Corp., Downington, Pa, USA). The lasso tool was used to outline the grey matter (GM) and white matter (WM) for each section and the threshold tool used to calculate the percentage of area stained in the GM and WM of each case.
Where required NRF-2 immunoreactivity was imaged using an Axio Lab.A1 microscope (Carl Zeiss, Jena, Germany). Sections were imaged using a 40x objective, and converted to JPEG files for analysis via the ZEN software (Carl Zeiss, Jena, Germany). Pixel counts were then determined using ImageJ software. The average percentage of area stained in the GM of each section was used for statistical analysis.
Western blot analysis
Western blots were performed on tissue homogenates as described previously59. Clarified supernatants were, diluted to matching protein concentration (23 μg total) and heated with Laemmli loading buffer (BioRad, Hercules, CA; 95 °C, 5 min) and separated on a 12% TGX gel (Bio-Rad, #161-0180) by SDS-PAGE. Prior to protein transfer to the blotting membrane all protein bands were visualized using a ChemiDocTM MP system (BioRad) and total band densities were obtained via Image Lab v5.2. Proteins were then transferred onto a PVDF membrane, blocked in 5% w/v skim milk powder (in 0.1% v/v TBS-T; 1 h, at 23 °C), then incubated with a mouse anti-Phospho-Tau (AT8; Thermo Scientific, Waltham, MA, #MN1020) overnight at 4 °C. Membranes were blocked at 4 °C and incubated with a rabbit anti-NRF2 antibody (Thermo Fisher Scientific, #710574) for 3 h, at 23 °C and then incubated with either an anti-mouse or anti-rabbit HRP-conjugated secondary antibody (dilution 1:5000 v/v; 1 h at 23 °C). Protein bands were visualized using Luminata Forte HRP substrate (Millipore, Billercia, USA) and imaged on a ChemiDocTM MP system (BioRad).
Direct Enzyme-Linked Immunosorbent Assay (ELISA)
Clarified supernatants (15 µg protein/mL; 100 µL) were prepared in coating buffer (containing 15 mM NaCO3 and 35 mM NaHCO3, pH 7.4), loaded onto a Maxisorp plate (Nalge Nunc, Rochester, USA) and incubated overnight at 4 °C. The plate was then blocked for 2 h in 1% w/v skim milk and incubated with primary antibody (3 h, 23 °C). Primary antibodies included goat anti-SOD1 (Santa Cruz, USA, #sc8637; dilution 1:200 v/v), goat anti-SOD2 (Sigma, USA, #S2147; 1:200 v/v) and goat anti-GPX1 (R&D Systems, USA, #AF3798; 1:500 v/v). Wells were then incubated with secondary antibody (1:5000 v/v dilution) (anti-mouse IgG-HRP and anti-rabbit IgG-HRP; 1 h at 20 °C). Next, a solution of ABTS substrate (Life Technologies, Carlsbad, CA) was added to each well followed by (1% w/v SDS) and monitoring at 410 nm using the FLUOstar Omega reader with values normalized to total protein.
Antioxidant activity assays
Total Superoxide dismutase (SOD) activity
The combined SOD1/2 (or total SOD) activity60 was assessed in samples of clarified supernatants (20 µL volume loaded) by measuring the inhibition of pyrogallol auto-oxidation monitored at 405 nm at 5 min intervals over 1 h of monitoring to establish a linear rate of superoxide radical anion evolution. Total SOD activity was expressed as a relative rate per min and normalized to the total protein level in the corresponding samples.
Total Glutathione peroxidase (GPX) activity
Assessment of total GPX activity was based on the GPX-dependent consumption of NADPH monitored at 340 nm61. Clarified supernatant (10 µL) were treated with a reaction mixture containing: 1 mM EDTA, 1 mM sodium azide, 1 mM glutathione, 1 U glutathione reductase, 0.25 mM NADPH and 50 mM phosphate-buffered saline (PBS) (volume 125 µL). A second mixture containing 350 µM H2O2 in 50 mM PBS (volume 65 µL) was then added to each well to a final volume of 200 µL. Absorbance was read at 5 min intervals over a 1 h period. Total GPX activity was expressed as a relative rate per min; all data was normalized to total protein in each sample.
Total Catalase (CAT) activity
To assess CAT activity, clarified supernatants (3.5 µL volume loaded) were treated with either 20 mM aminotriazole (AT) (a dose that inhibits CAT activity62,63, the heme-poison sodium azide (SA) (20 mM) as an additional negative control or 50 mM PBS (as vehicle control) for 5 min at 20 °C. Next, an aliquot (95 µL) of 0.1% v/v H2O2 was added and absorbance was read at 240 nm using a FLUOstar Omega reader at 5 min intervals over 2 h. The rate of H2O2 decomposition was assessed by the gradient of the linear data analysis (coefficient R2 ranging 0.9–0.95) and normalized to total protein in the corresponding samples with CAT activity ascribed specifically to an AT-inhibited fraction of peroxide consumption.
Total Peroxiredoxin (Prx) activity
A modified ferrous oxidation-xylenol orange (FOX) assay was used to assess total Prx activity as described previously64. Cumene hydroperoxide was selected to enhance specificity in this enzyme activity assay as Prx activity shows enhanced catalytic efficacies and specificities toward tertiary lipid hydroperoxides65. Samples of clarified supernatants (3.5 µL) were incubated with 100 µM cumene hydroperoxide and 100 µM dithiothreitol (final volume of 10 µL) for 5 min at 20 °C. An aliquot of FOX solution (250 µM ammonium sulfate, 100 mM D-sorbitol and 125 µM xylenol orange) was then added to yield a total volume of 100 µL and the mixture was incubated at 20 °C. After 30 min, absorbance was read at 560 nm using a FLUOstar Omega reader and data was normalized to total protein.
Total hemeoxygenase (HO) activity
Clarified supernatants from brain homogenates (500 μL) were subject to ultracentrifigation (100 000 × g; 1 h at 4 °C). The resulting microsomal pellets were resuspended in 100 µL PBS and used to determine total heme-oxygenase (HO 1/2) protein activity. For total HO activity microsomes were mixed at a final ratio f 2:1 (brain homogenate-microsomes/isolated at liver-microsomes) in 100 µL buffer (250 mM sucrose, 20 M Tris, pH 7.4). Next, the microsomal mixtures were treated with 1 mM NADPH, 2 mM D-glucose-6-phosphate (G-6-P), 1U glucose-6-phosphate dehydrogenase and 1 µL of 2.5 mmol/L hemin and incubated at 37 °C in the dark. After 1 h, the reaction was stopped by addition of 100 µL ethanol/DMSO (95:5 v/v). The mixture was then centrifuged at 13000 × g for 5 min, and the supernatant was assayed for bilirubin with liquid chromatography20. Finally, total HO activity was determined as the protein normalized level of bilirubin.
Caspase activation
Caspase-3 and -7 activities were assessed using a commercial kit (Caspase-GloTM 3/7, Promega, Fitchburg, USA) as per the manufacturer’s instructions. Briefly clarified supernatants (50 μL) were diluted according to individual protein concentrations to yield final protein concentrations of 0.5 mg protein/mL. These protein-adjusted samples were then added to Caspase-GloTM 3/7 reagent (1:1 v/v, 20 °C for 60 min) and luminescence measured using a FLUOstar Omega reader (BMG LABTECH, Offenburg, Germany). Total caspase 3/7 activity was represented as mean luminescence after 60 min of monitoring.
Statistical analysis
All data are presented as the mean ± SD. Statistical tests including two-tailed t-tests (for paired data sets), and ANOVA were performed with GraphPad Prism (v6); p ≤ 0.05 was regarded as significant.
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request. All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
References
- 1.Halliday, G. M., Double, K. L., Macdonald, V. & Kril, J. J. Identifying severely atrophic cortical subregions in Alzheimer’s disease. Neurobiology of aging24, 797–806 (2003).
- 2.West, M. J., Kawas, C. H., Martin, L. J. & Troncoso, J. C. The CA1 region of the human hippocampus is a hot spot in Alzheimer’s disease. Annals of the New York Academy of Sciences908, 255–259 (2000).
- 3.Braak, H. & Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol (Berl)82, 239–259 (1991).
- 4.Newell, K. L., Hyman, B. T., Growdon, J. H. & Hedley-Whyte, E. T. Application of the National Institute on Aging (NIA)-Reagan Institute criteria for the neuropathological diagnosis of Alzheimer disease. J Neuropathol Exp Neurol58, 1147–1155 (1999).
- 5.Gomez-Isla, T. et al. Profound loss of layer II entorhinal cortex neurons occurs in very mild Alzheimer’s disease. The Journal of neuroscience: the official journal of the Society for Neuroscience16, 4491–4500 (1996).
- 6.Hardy, J. & Selkoe, D. J. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science297, 353–356 (2002).
- 7.Sutherland, G. T., Siebert, G. A., Kril, J. J. & Mellick, G. D. Knowing me, knowing you: can a knowledge of risk factors for Alzheimer’s disease prove useful in understanding the pathogenesis of Parkinson’s disease? J Alzheimers Dis25, 395–415 (2011).
- 8.Nunomura, A. et al. Oxidative damage to RNA in neurodegenerative diseases. Journal of biomedicine & biotechnology2006, 82323 (2006).
- 9.Sultana, R. & Butterfield, D. A. Role of oxidative stress in the progression of Alzheimer’s disease. J Alzheimers Dis19, 341–353 (2010).
- 10.Butterfield, D. A., Swomley, A. M. & Sultana, R. Amyloid beta-peptide (1–42)-induced oxidative stress in Alzheimer disease: importance in disease pathogenesis and progression. Antioxidants & redox signaling19, 823–835 (2013).
- 11.De Felice, F. G. et al. Abeta oligomers induce neuronal oxidative stress through an N-methyl-D-aspartate receptor-dependent mechanism that is blocked by the Alzheimer drug memantine. The Journal of biological chemistry282, 11590–11601 (2007).
- 12.Nunomura, A. et al. Oxidative damage is the earliest event in Alzheimer disease. J Neuropathol Exp Neurol60, 759–767 (2001).
- 13.Cuajungco, M. P. et al. Evidence that the beta-amyloid plaques of Alzheimer’s disease represent the redox-silencing and entombment of abeta by zinc. The Journal of biological chemistry275, 19439–19442 (2000).
- 14.Wataya, T. et al. High molecular weight neurofilament proteins are physiological substrates of adduction by the lipid peroxidation product hydroxynonenal. The Journal of biological chemistry277, 4644–4648 (2002).
- 15.Sutherland, G. T., Chami, B., Youssef, P. & Witting, P. K. Oxidative stress in Alzheimer’s disease: Primary villain or physiological by-product? Redox report: communications in free radical research18, 134–141 (2013).
- 16.Retz, W., Gsell, W., Munch, G., Rosler, M. & Riederer, P. Free radicals in Alzheimer’s disease. Journal of neural transmission. Supplementum54, 221–236 (1998).
- 17.Markesbery, W. R. Oxidative stress hypothesis in Alzheimer’s disease. Free radical biology & medicine23, 134–147 (1997).
- 18.Musiek, E. S. & Holtzman, D. M. Three dimensions of the amyloid hypothesis: time, space and ‘wingmen’. Nature neuroscience18, 800–806 (2015).
- 19.Mills, J. D. et al. The alternative splicing of the apolipoprotein E gene is unperturbed in the brains of Alzheimer’s disease patients. Molecular biology reports41, 6365–6376 (2014).
- 20.Thomas, S. R., Witting, P. K. & Stocker, R. 3-Hydroxyanthranilic acid is an efficient, cell-derived co-antioxidant for alpha-tocopherol, inhibiting human low density lipoprotein and plasma lipid peroxidation. The Journal of biological chemistry271, 32714–32721 (1996).
- 21.Johnston, M. A. & Delwiche, E. A. Distribution and Characteristics of The Catalases of Lactobacillaceae. Journal of bacteriology90, 347–351 (1965).
- 22.Kim, S. H., Fountoulakis, M., Cairns, N. & Lubec, G. Protein levels of human peroxiredoxin subtypes in brains of patients with Alzheimer’s disease and Down syndrome. Journal of neural transmission. Supplementum, 223–235 (2001).
- 23.Lau, A., Tian, W., Whitman, S. A. & Zhang, D. D. The predicted molecular weight of Nrf2: it is what it is not. Antioxidants & redox signaling18, 91–93 (2013).
- 24.Sutherland, G. T., Janitz, M. & Kril, J. J. Understanding the pathogenesis of Alzheimer’s disease: will RNA-Seq realize the promise of transcriptomics? J Neurochem116, 937–946 (2011).
- 25.Selkoe, D. J. & Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO molecular medicine8, 595–608 (2016).
- 26.Tramutola, A., Lanzillotta, C., Perluigi, M. & Butterfield, D. A. Oxidative stress, protein modification and Alzheimer disease. Brain research bulletin133, 88–96 (2017).
- 27.Greenough, M. A., Camakaris, J. & Bush, A. I. Metal dyshomeostasis and oxidative stress in Alzheimer’s disease. Neurochemistry international62, 540–555 (2013).
- 28.Valko, M., Jomova, K., Rhodes, C. J., Kuca, K. & Musilek, K. Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Archives of toxicology90, 1–37 (2016).
- 29.Gsell, W. et al. Decreased catalase activity but unchanged superoxide dismutase activity in brains of patients with dementia of Alzheimer type. J Neurochem64, 1216–1223 (1995).
- 30.Thome, J. et al. Oxidative-stress associated parameters (lactoferrin, superoxide dismutases) in serum of patients with Alzheimer’s disease. Life sciences60, 13–19 (1997).
- 31.Chang, Y.-T. et al. The Roles of Biomarkers of Oxidative Stress and Antioxidant in Alzheimer’s Disease: A Systematic Review. BioMed Research International2014, 182303 (2014).
- 32.Cristalli, D. O., Arnal, N., Marra, F. A., de Alaniz, M. J. & Marra, C. A. Peripheral markers in neurodegenerative patients and their first-degree relatives. Journal of the neurological sciences314, 48–56 (2012).
- 33.Grillo, F. et al. Factors affecting immunoreactivity in long-term storage of formalin-fixed paraffin-embedded tissue sections. Histochemistry and cell biology144, 93–99 (2015).
- 34.Surh, Y. J., Kundu, J. K. & Na, H. K. Nrf2 as a master redox switch in turning on the cellular signaling involved in the induction of cytoprotective genes by some chemopreventive phytochemicals. Planta medica74, 1526–1539 (2008).
- 35.Ramsey, C. P. et al. Expression of Nrf2 in neurodegenerative diseases. J Neuropathol Exp Neurol66, 75–85 (2007).
- 36.Rojo, A. I., Sagarra, M. R. & Cuadrado, A. GSK-3beta down-regulates the transcription factor Nrf2 after oxidant damage: relevance to exposure of neuronal cells to oxidative stress. J Neurochem105, 192–202 (2008).
- 37.Benchekroun, M. et al. The Antioxidant Additive Approach for Alzheimer’s Disease Therapy: New Ferulic (Lipoic) Acid Plus Melatonin Modified Tacrines as Cholinesterases Inhibitors, Direct Antioxidants, and Nuclear Factor (Erythroid-Derived 2)-Like 2 Activators. Journal of medicinal chemistry59, 9967–9973 (2016).
- 38.Buendia, I. et al. New melatonin-cinnamate hybrids as multi-target drugs for neurodegenerative diseases: Nrf2-induction, antioxidant effect and neuroprotection. Future medicinal chemistry7, 1961–1969 (2015).
- 39.Barone, E. et al. Heme oxygenase-1 posttranslational modifications in the brain of subjects with Alzheimer disease and mild cognitive impairment. Free radical biology & medicine52, 2292–2301 (2012).
- 40.Barone, E. et al. Biliverdin reductase–a protein levels and activity in the brains of subjects with Alzheimer disease and mild cognitive impairment. Biochimica et biophysica acta1812, 480–487 (2011).
- 41.Barone, E. et al. Impairment of biliverdin reductase-A promotes brain insulin resistance in Alzheimer disease: A new paradigm. Free radical biology & medicine91, 127–142 (2016).
- 42.Stocker, R., Yamamoto, Y., McDonagh, A. F., Glazer, A. N. & Ames, B. N. Bilirubin is an antioxidant of possible physiological importance. Science235, 1043–1046 (1987).
- 43.Stocker, R., Glazer, A. N. & Ames, B. N. Antioxidant activity of albumin-bound bilirubin. Proceedings of the National Academy of Sciences of the United States of America84, 5918–5922 (1987).
- 44.He, M. et al. Heme oxygenase-1-derived bilirubin protects endothelial cells against high glucose-induced damage. Free radical biology & medicine89, 91–98 (2015).
- 45.Chen, W. et al. Absence of the biliverdin reductase-a gene is associated with increased endogenous oxidative stress. Free radical biology & medicine115, 156–165 (2017).
- 46.Stifter, J. et al. Neuroprotection and neuroregeneration of retinal ganglion cells after intravitreal carbon monoxide release. PloS one12, e0188444 (2017).
- 47.Jazwa, A. & Cuadrado, A. Targeting heme oxygenase-1 for neuroprotection and neuroinflammation in neurodegenerative diseases. Current drug targets11, 1517–1531 (2010).
- 48.Massaad, C. A., Washington, T. M., Pautler, R. G. & Klann, E. Overexpression of SOD-2 reduces hippocampal superoxide and prevents memory deficits in a mouse model of Alzheimer’s disease. Proceedings of the National Academy of Sciences of the United States of America106, 13576–13581 (2009).
- 49.Dumont, M. et al. Reduction of oxidative stress, amyloid deposition, and memory deficit by manganese superoxide dismutase overexpression in a transgenic mouse model of Alzheimer’s disease. The FASEB Journal23, 2459–2466 (2009).
- 50.Melov, S. et al. Mitochondrial oxidative stress causes hyperphosphorylation of tau. PloS one2, e536 (2007).
- 51.Esposito, L. et al. Reduction in mitochondrial superoxide dismutase modulates Alzheimer’s disease-like pathology and accelerates the onset of behavioral changes in human amyloid precursor protein transgenic mice. The Journal of neuroscience: the official journal of the Society for Neuroscience26, 5167–5179 (2006).
- 52.Crack, P. J., Cimdins, K., Ali, U., Hertzog, P. J. & Iannello, R. C. Lack of glutathione peroxidase-1 exacerbates Abeta-mediated neurotoxicity in cortical neurons. Journal of neural transmission (Vienna, Austria: 1996)113, 645–657 (2006).
- 53.Kanninen, K. et al. Nuclear factor erythroid 2-related factor 2 protects against beta amyloid. Molecular and cellular neurosciences39, 302–313 (2008).
- 54.Amin, F. U., Shah, S. A. & Kim, M. O. Vanillic acid attenuates Aβ(1-42)-induced oxidative stress and cognitive impairment in mice. Scientific Reports7, 40753 (2017).
- 55.Montine, T. J., Sonnen, J. A., Montine, K. S., Crane, P. K. & Larson, E. B. Adult Changes in Thought study: dementia is an individually varying convergent syndrome with prevalent clinically silent diseases that may be modified by some commonly used therapeutics. Current Alzheimer research9, 718–723 (2012).
- 56.Groebler, L. K. et al. Cosupplementation with a synthetic, lipid-soluble polyphenol and vitamin C inhibits oxidative damage and improves vascular function yet does not inhibit acute renal injury in an animal model of rhabdomyolysis. Free radical biology & medicine52, 1918–1928 (2012).
- 57.Bustin, S. A. et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clinical chemistry55, 611–622 (2009).
- 58.Esterbauer, H., Schaur, R. J. & Zollner, H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free radical biology & medicine11, 81–128 (1991).
- 59.Antao, S. T., Duong, T. T., Aran, R. & Witting, P. K. Neuroglobin overexpression in cultured human neuronal cells protects against hydrogen peroxide insult via activating phosphoinositide-3 kinase and opening the mitochondrial K(ATP) channel. Antioxidants & redox signaling13, 769–781 (2010).
- 60.Parry, S. N., Ellis, N., Li, Z., Maitz, P. & Witting, P. K. Myoglobin induces oxidative stress and decreases endocytosis and monolayer permissiveness in cultured kidney epithelial cells without affecting viability. Kidney & blood pressure research31, 16–28 (2008).
- 61.Wilson, S. R., Zucker, P. A., Huang, R. R. C. & Spector, A. Development of synthetic compounds with glutathione peroxidase activity. Journal of the American Chemical Society111, 5936–5939 (1989).
- 62.Dao, V. T. et al. Catalase activity prevents exercise-induced up-regulation of vasoprotective proteins in venous tissue. Journal of cellular and molecular medicine15, 2326–2334 (2011).
- 63.Ma, H. P. Hydrogen peroxide stimulates the epithelial sodium channel through a phosphatidylinositide 3-kinase-dependent pathway. The Journal of biological chemistry286, 32444–32453 (2011).
- 64.Nelson, K.J. & Parsonage, D. Measurement of peroxiredoxin activity. Current protocols in toxicology/editorial board, Mahin D. Maines Chapter 7, Unit710 (2011).
- 65.Parsonage, D., Karplus, P. A. & Poole, L. B. Substrate specificity and redox potential of AhpC, a bacterial peroxiredoxin. Proceedings of the National Academy of Sciences of the United States of America105, 8209–8214 (2008).
Acknowledgements
The authors would firstly like to thank the donors and their families for their kind gift that has allowed this research to be undertaken. A special thanks to A/Prof David Sullivan and Terrance Foo from Department of Clinical Biochemistry, Royal Prince Alfred Hospital, Sydney, for the APOE genotyping. We also thank the New South Wales Brain Banks for providing tissue samples and assistance with clinical and pathological data. The NSWBB is made up of the Sydney Brain Banks (SBB) and NSW Tissue Resource Centre (NSW TRC). The SBB is supported by Neuroscience Research Australia, the University of New South Wales, the National Health and Medical Research Council (NHMRC) and the Australian Brain Bank Network. NSW BTRC is supported by the University of Sydney, NHMRC, the Schizophrenia Research Institute and the National Institutes of Alcoholism and Alcohol Abuse (NIAAA; (R24 AA012725). This work was supported by an ARC grant (DP130103711 to P.K.W.) and the Judith Jane Mason and Harold Stannett Williams Memorial Foundation GIA (awarded to G.S. and P.K.W.). P.Y. acknowledges the support of the Sydney Medical School Bluesand Foundation Scholarship that provided her stipend to undertake her PhD studies.
Author information
P.Y. performed the bulk of the experimental work, data analysis and contributed to construction of the manuscript. B.C., J.L. and T.M. contributed to collaborative experimental data. G.S. designed and tested the specificity of primers for monitoring gene regulation. G.S. and P.K.W. assisted in developing research direction based on experimental outcomes, data analysis and contributed to the preparation of the manuscript. Together G.S. and P.K.W. conceived the study.
Correspondence to Paul K. Witting.
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Received
Accepted
Published
DOI
Further reading
Kaempferide prevents cognitive decline via attenuation of oxidative stress and enhancement of brain‐derived neurotrophic factor/tropomyosin receptor kinase B/cAMP response element‐binding signaling pathway
Phytotherapy Research (2019)Protective Effects of Pyruvic Acid Salt Against Lithium Toxicity and Oxidative Damage in Human Blood Mononuclear Cells
Advanced Pharmaceutical Bulletin (2019)Crosstalk between Oxidative Stress and Tauopathy
International Journal of Molecular Sciences (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Abstract
Commercial superoxide dismutase [SOD] is derived from melon extract and has a potential as a dietary supplement due to its beneficial antioxidative effects. We aimed to improve the productivity of SOD compared with plant SOD by using a generally regarded as safe [GRAS] microorganism, Bacillus amyloliquefaciens, and assess its antioxidative effect using γ-radiation- and dextransulphate sodium [DSS]-induced oxidative models in mice.
Methods
We identified the sodA gene encoding manganese-containing SODs [Mn-SOD] in B. amyloliquefaciens, constructed a Mn-SOD deficient mutant, and screened a high-SOD-producing strain. We compared the antioxidative effect of orally administered enteric-coated SOD protein partially purified from B. amyloliquefaciens with wild-type and high-SOD-producing strain spores. The effect of SOD on DSS-induced colitis was also investigated. Colonic inflammation was assessed using disease activity index, macroscopic and histological damage scores, antioxidant enzyme activities, and inflammatory cytokines.
The SOD activity of B. amyloliquefaciens is derived from secreted Mn-SOD encoded by the sodA gene, as shown by comparing sodA knock-out mutant spores with wild-type and high-SOD-producing spores. Enteric-coated SOD of B. amyloliquefaciens appears to be effective in reducing oxidative stress in γ-radiation- and DSS-induced mouse models. Co-administration of SOD with wild-type B. amyloliquefaciens or high-SOD-producer strain spores showed a synergistic effect. SOD enzyme and B. amyloliquefaciens spores contribute to the reduction of oxidative stress and inflammatory response in DSS-induced colitis.
Conclusions
Mn-SOD of B. amyloliquefaciens could be another source of SOD supplement and may be useful to prevent and treat ulcerative colitis.
Inflammatory bowel diseases, superoxide dismutase, Bacillus amyloliquefaciens
1. Introduction
Oxidative stress, involved in many diseases, is defined as an impaired balance between reactive oxygen species [ROS] production and antioxidant defences. Antioxidant enzymes, such as superoxide dismutase [SOD], play a key role in diminishing oxidative stress.1,2 Thus, the removal of ROS by exogenous SODs has been suggested as an effective preventive strategy against various diseases, and oral supplementation with melon-derived SOD has been shown to have a therapeutic value in both animal and clinical trials.1,3 SODs are widely distributed in prokaryotic and eukaryotic cells, and have been classified into four families based on their different types of metal centres [copper/zinc, nickel, manganese, and iron].4 Manganese-containing SODs [Mn-SODs] are widely present in many bacteria, chloroplasts, mitochondria, and cytosol of eukaryotic cells. Mn-SODs in the mitochondria have been suggested to function as tumour suppressors, and cancer cells generally have diminished Mn-SOD activity.5
Aerobic organisms generate ROS, and environmental stresses induced by a wide range of environmental factors, including heavy metals, can lead to overproduction of ROS and alter intracellular redox status, ultimately leading to cell death.6 It has been reported that the heavy metal resistance of several Bacillus strains is related to oxidative enzyme activities.6,7 The effect of probiotic Bacillus coagulans in alleviating mercury toxicity has also been reported in rats.8 Ionising radiation exposure causes direct ionisation of cellular atoms and secondary activation, in which energy is transferred to atomic particles, such as electrons, that may lead to changes in cellular function and most commonly activation of cells. Exogenous SOD has been shown to have the ability to repair vegetative B. subtilis cell damage and improve cell survival rate and internal SOD activity in cells irradiated by gamma radiation.9 A diet containing B. subtilis strain increased the SOD level of serum or tissues significantly in several fish strains.10–13B. subtilis has been found to increase the level of antioxidant enzymes, including SOD, in broiler chickens.14 These findings led us to develop Bacillus SOD as an alternative antioxidative enzyme for oral supplementation. The microbial fermentation could be extended in capacity relatively easily, and productivity could be maximised through the breeding of the strain and optimisation of fermentation.
B. amyloliquefaciens, recently determined to be a separate species from B. subtilis, produces spores with strong resistance to adverse conditions, making it well-suited for commercial use.15 In 1999, the Food and Drug Administration [FDA] published a final rule in the Federal Register affirming that carbohydrase and protease enzyme preparations from B. subtilis or B. amyloliquefaciens are generally regarded as safe [GRAS] for use as direct food ingredients.16 The species traditionally included in the B. subtilis group [B. subtilis, B. licheniformis, B. amyloliquefaciens, and B. pumilus] have also been given a ‘Qualified Presumption of Safety’ by the European Food Safety Authority.17
Many studies have reported on the properties of B. amyloliquefaciens, including its antioxidant effects and probiotic potential.18–25B. amyloliquefaciens has also been reported to be useful in the management of inflammatory bowel disease [IBD].26 However, there are no studies to date regarding the use of any SOD enzyme from Bacillus strains, including B. amyloliquefaciens. Therefore, we tested B. amyloliquefaciens strain GF423 as a SOD producer.
B. amyloliquefaciens strain GF423, formerly classified as B. polyfermenticus, belongs to the species B. amyloliquefaciens according to molecular taxonomy. It is more closely related to B. amyloliquefaciens FZB42, the type strain of B. amyloliquefaciens subspecies plantarum, than to B. amyloliquefaciens DSM7, the type strain of B. amyloliquefaciens subspecies amyloliquefaciens. Its ability to produce more SOD than wild-type B. amyloliquefaciens renders it an excellent candidate for SOD production for the treatment of inflammatory bowel disease [IBD].
IBD is a chronic inflammatory disorder of the gastrointestinal tract. Crohn’s disease and ulcerative colitis, the two most common clinical forms of IBD, affect millions of patients around the world.27,28 Oxidative stress plays a pivotal role in the initiation and progression of IBD.29 Decreased levels of SOD activity are associated with increased inflammatory processes, indicating that inflammation may be caused by insufficient antioxidant activity in patients with IBD.30,31 Since the local colonic delivery of SOD by bacteria is effective in reducing experimental colitis, and lactic acid bacteria [LAB] have been used as vehicles to deliver therapeutic agents in the gut, engineering LAB to produce SOD has been investigated as a supplement to traditional IBD treatments.32–36 However, the use of genetically modified bacteria in humans remains difficult to date; therefore, the use of wild-type bacteria selected for their production of SOD, and/or other enzymes that neutralise ROS, may be a more promising means of IBD treatment.37 Until now, no research regarding oral administration of SOD for IBD treatment has been tried, due to the assumption of its inefficient delivery because of the enzyme’s short half-life. However, enteric coatings may make the oral delivery of SOD feasible.
In this work, we identified the Mn-SOD gene of B. amyloliquefaciens, purified the SOD enzyme, and screened a high-SOD-producing strain for the production of SOD on an industrial scale. We also assessed the possibility of enteric-coated SOD as a dietary supplement using γ-radiation- and DSS-induced ulcerative colitis mouse models.
2. Materials and Methods
2.1. Chemical compounds used
Dextran sulphate sodium [DSS] was obtained from MP Biochemicals Korea [Songpa-gu, Seoul, Korea]. The enzyme assay kit, for measuring superoxide dismutase [SOD], catalase [CAT] ,and glutathione peroxidase [GPx] levels, was purchased from Cayman Chemical Company [Ann Arbor, MI, USA]. Enzyme-linked immunosorbent assay [ELISA] kits, for measuring inflammatory cytokines tumour necrosis factor alpha [TNF-α], interleukin 1 beta [IL1-β], and interleukin 6 [IL-6], were obtained from R&D systems [Minneapolis, MN, USA].
2.2. Bacterial strains and DNA manipulation
B. amyloliquefaciens GF423 was obtained from the Korean Collection for Type Cultures. The sodA gene was cloned by polymerase chain reaction [PCR] from the genome of B. amyloliquefaciens GF423 using the following primers based on DNA sequence of B. amyloliquefaciens FZB42: 5′ gggatgaacacaagtgagag 3′ and 5′ aagctcatgaccacagcaag 3′. The PCR product of about 2 kb was cloned using Mighty TA-cloning Kit [Takara] and its nucleotide sequence was determined by ABI3730XL DNA analyser [Applied Biosystems]. The strain with deletion of sodA function, B. amyloliquefaciens ΔsodA, was constructed by a double-crossover recombination as follows. A pair of DNA fragments flanking the sodA gene of B. amyloliquefaciens GF423 for homologous recombination was amplified using PCR with the following primers: 5′ aaacagctgggatgaacacaagtgagag 3′ and 5′ cacactcttaagtttgcttccaattctggaagtttgtaag 3′ for the upstream fragment; and 5′ ctactgacagcttccaaggatacctgaactaccaaaaccg 3′ and 5′ aaacagctgaagctcatgaccacagcaag 3′ for the downstream fragment. The erythromycin-resistant [EmR] gene was amplified using PCR from pDG166438 with the following primers: 5′ gaagcaaacttaagagtgtg 3′ and 5′ tccttggaagctgtcagtag 3′. The upstream fragment, the EmR gene, and downstream fragment were assembled in order and cloned into the PvuII site of plasmid pUori-ts containing the temperature-sensitive replication origin working in B. subtilis.39 The resulting plasmid was transformed into B. amyloliquefaciens GF423 by electroporation.40 The sodA knock-out mutant was selected from the transformants resistant to Em [5 μg/mL] at 42 °C and then plasmid-cured at 30 °C. The genomic status around the sodA region was confirmed by PCR and DNA sequencing of the PCR product. The sodA overproducing strain, B. amyloliquefaciens GF424, was selected from a mutant library, which was prepared using ultraviolet [UV] irradiation.41 The sodA gene of B. amyloliquefaciens GF424 was confirmed to be identical with that of the wild type by DNA sequencing. B. amyloliquefaciens strains were cultivated in tryptic soy broth at 37°C [BD]. PCR was performed using standard methods with Takara Advantage 2 Polymerase. Restriction enzymes and DNA-modifying enzymes were purchased from New England Biolabs [NEB]. All other reagents were purchased from Sigma unless otherwise mentioned.
2.3. Preparation of test materials
The test materials for oral administration were prepared as follows. For preparation of SOD powder, B. amyloliquefaciens GF424 was grown in tryptic soy broth at 37°C for 24 h. The culture supernatant was collected by centrifugation at 7500 rpm for 10 min at 4°C. The collected supernatant was filtered through 0.25-μM pore-size membrane [Sartorius] and then concentrated 10-fold by ultrafiltration using a polyethersulphone membrane [Sartorius] with a molecular weight cut-off of 10 kDa. Enteric coating was conducted as follows: UF concentrate was mixed with the same volume of 0.1% [w/v] shellac solution dissolved in distilled water and 5% ethanol, and the mixture was lyophilised. For the preparation of the endospore, B. amyloliquefaciens strains were grown in tryptic soy broth at 37°C for about 48 h. The precipitate obtained by centrifugation at 7500 rpm for 10 min at 4°C was washed with phosphare-buffered saline [PBS] solution [Sigma], and the endospores were purified from the precipitate using the method described by Tavares et al.42
2.4. Purification and protein analysis
An 800-ml culture supernatant of B. amyloliquefaciens GF423 was collected by centrifugation, and filtered and concentrated as described above. Protamine sulphate was added to the concentrate to reach 0.2% [w/v], followed by centrifugation at 12000 rpm for 45 min at 4°C. The supernatant obtained after centrifugation was precipitated by adding ammonium sulphate up to 60% [w/v] and stirring for 30 min on ice. The supernatant obtained from ammonium sulphate precipitation was applied onto a Phenyl Sepharose HP column [16 × 10 cm] [GE Healthcare Life Sciences, USA] equilibrated with 50-mM potassium phosphate buffer [pH 7.5]. Proteins were eluted with a linear gradient of 2 to 0 M ammonium sulphate equilibrated in 50-mM potassium phosphate buffer. The fractions containing SOD activity were pooled and concentrated with Centricon [Merck, USA] with molecular weight cut-off [MWCO] of 10 kd. The concentrate was dialysed against 50 mM-potassium phosphate buffer [pH 7.5]. Enzyme purity was determined by SDS-PAGE analysis. Protein was quantitated using Bradford protein assay kit [BioRad, USA]. The N-terminal sequence was determined with a model 492 protein sequencer [Applied Biosystems, USA].
2.5. Experimental animals and diet
Six-week-old female Balb/c mice weighing 16–20 g were purchased from Orient Bio [Seongnam, Korea]. All mice were housed in individual cages under pathogen-free and constant conditions [temperature 25 ± 2 °C; humidity 70-75%, lighting regimen 12-h light/12-h dark]. Mice were fed pellet diet and tapwater. The study was approved by the Korea Research Institute of Bioscience and Biotechnology Review Board for the care of animals.
2.6. Oxidative stress model and SOD treatment
The mice were randomly separated into nine groups [n = 7]. The duration of administration was 28 days in all cases. All spores were administered at a rate of 107/day and all BA SOD at a rate of 10 U/day in 100 µl PBS. The groups included: the control group, which was orally supplemented with 100 µl phosphate-buffered solution [PBS]; the B. amyloliquefaciens SOD [BA SOD] group, which was administered wild-type BASOD; the oxidative stress [OS] control group, supplemented with 100 µl PBS and irradiated with γ-ray 2Gy every 7days; the OS BASOD group, which was administered wild-type BASOD and irradiated with γ-rays at a dose of 2Gy every 7 days; the wild-type B. amyloliquefaciens [BA WT] spore group; the B. amyloliquefaciens sodA knock-out mutant [BA ΔSODA] spore group; the B. amyloliquefaciens high-SOD-producing [BA HPSOD] spore group; the BA WT spore+ BA SOD group, administered full doses of BA WT spore and BA SOD; and the BA HP SOD spore + BA SOD group, administered full doses of BA HP SOD spore and BA SOD. Blood was collected on the first and last days, incubated for 30 min at 25°C, and centrifuged at 1300 rpm for 30 min at 4°C. Supernatants were transferred to a new e-tube and stored at -80°C deep freezer.
2.7. DSS-induced IBD and treatment
The mice were divided into eight groups [n = 7]. All spores were administered at a rate of 107/day in 100 µl PBS for 3 weeks, and all DSS was provided as 3% DSS in drinking water for the last 2 weeks. Groups included: the control group, which was administered 100 µl PBS for 3 weeks; the DSS group, administered 100 µl PBS for 3 weeks and provided DSS; the BA WT spore group, administered BA WT spore and provided DSS; the BA ΔSOD spore group, administered BA ΔSOD spore and provided DSS; the BA HP SOD spore group, administered BA HP SOD spore and provided DSS; the BA WT spore + BA SOD group, administered full doses BA WT SOD spore and BA SOD and provided DSS; the BA HP SOD spore + BA SOD group, administered full doses of BA HP SOD spore and BA SOD 10 and provided DSS; and the BA SOD group, administered BA SOD and provided DSS. Blood was collected and tested as described above.
2.8. Measurement of the activity index of IBD
The activity index of IBD was evaluated by an independent observer. IBD activity indexes included weight loss, stool consistency, mean score of rectal bleeding, and length of colon. Each score was determined as follows: consistency of stools [score: 0, well-formed pellet; 1, smooth, semi-moulded but not attached to the anus; 2, semi-muolded but attached to the anus; 3, liquid and liquid faeces], rectal bleeding [score: 0, normal; 1, blood traces in rectum; 2, blood in whole rectum; 3, blood traces in both rectum and stool; 4, total rectal bleeding], and weight loss rate calculated as a percentage of baseline body weight [body weight at Day 0].
2.9. Measurement of SOD, CAT, and GPx activities
The SOD, CAT, and GPx activities in serum samples were measured according to the manual provided by suppliers of the kits [Cayman Chemical Company, Ann Arbor, MI, USA].
2.10. Histology
The colon tissues were fixed in 4% formaldehyde, paraffin-embedded, cleared with xylene, hydrated, and stained with haematoxylin and eosin [H&E]. H&E-stained colonic tissue sections were scored by a blinded observer using a previously published system, for the following measures to grade the extent of inflammatory infiltrate [0–5], crypt damage [0–4], and ulceration [0–3], and the presence or absence ofo edema [0 or 1]. The scores represented the histological damage associated with DSS treatment in the mid-colon [from the distal end of the plicae to the two-thirds from the anus], where inflammation was the most severe.43
2.11. Measurement of inflammatory cytokines using ELISA
The levels of inflammatory cytokines IL-1β, TNF-α, and IL-6, were determined using commercial immunoassay kits in accordance with the manufacturer’s instructions [R&D systems, Minneapolis, MN, USA].
2.12. Statistical analysis
Analyses were performed using Microsoft Excel 2010 for Windows. Quantitative data are shown as mean ± SD. Significance was determined using a two-tailed, unpaired Student’s t-test; p-values < 0.05 were considered significant.
3. Results
3.1. Identification of the sodA gene encoding Mn-SOD in B. amyloliquefaciens
SOD activity was found in the culture supernatant of B. amyloliquefaciens GF423, which was expected to be attributable to a gene homologous with B. subtilis sodA encoding an extracellular SOD. We determined the N-terminal amino acid sequence of a protein with a molecular weight [MW] of about 22000 Da based on SDS PAGE observed in the culture supernatant of the strain. The N-terminal sequence, AYKLPE, was searched against the National Center for Biotechnology Information [NCBI] database using BLASTP, and found to match exactly with the N-terminal sequence of SOD derived from various B. amyloliquefaciens strains, which strongly suggests that the protein with an MW of 22000 Da was responsible for the SOD activity in the culture supernatant. We cloned the gene from B. amyloliquefaciens GF423 using PCR with primers corresponding to the flaking sequences of the sodA gene of B. amyloliquefaciens FZB42 [NC_009725], and determined its nucleotide sequence. The sodA gene of B. amyloliquefaciens GF423 encodes a protein of 201 amino acids with an MW of 22488 Da, which have the same N-terminal sequence as the protein in the culture supernatant. The amino acid sequence of SodA of B. amyloliquefaciens GF423 is completely identical with that of B. amyloliquefaciens FZB42, and shares 92% identity and 97% similarity with those of B. subtilis 168 [Figure 1].
Comparison of the amino acid sequence of SodA derived from B. amyloliquefaciens GF423 with those from B. amyloliquefaciens FZB42 and B. subtilis 168. BA_GF423 indicates B. amyloliquefaciens GF423, BA_FZB42; B. amyloliquefaciens FZB42; and BS_168, B. subtilis 168. Active sites of B. subtilis SodA5 are indicated by dots under the sequence.
Comparison of the amino acid sequence of SodA derived from B. amyloliquefaciens GF423 with those from B. amyloliquefaciens FZB42 and B. subtilis 168. BA_GF423 indicates B. amyloliquefaciens GF423, BA_FZB42; B. amyloliquefaciens FZB42; and BS_168, B. subtilis 168. Active sites of B. subtilis SodA5 are indicated by dots under the sequence.
In B. subtilis, the sodA gene encodes an extracellular Mn-SOD; together with a functional domain analysis by InterPro,44 this indicates that SodA of B. amyloliquefaciens GF423 is an extracellular Mn-SOD. Bacteria of the B. subtilis group, including B. amyloliquefaciens, have multiple sodA genes. For example, in B. subtilis and B. amyloliquefaciens, there are three types of genes encoding SOD: sodA, sodC, and sodF. Thus, to determine whether the SodA of B. amyloliquefaciens GF423 is a major determinant of extracellular SOD activity, the sodA gene was knocked out from its genome as described in the Materials and Methods section, and SOD activity was measured in the culture supernatant of the resulting knock-out mutant. SOD activity was not detected in the sodA knock-out mutant, which suggests that SodA is a major extracellular SOD in B. amyloliquefaciens GF423. This finding corroborates with previous reports that SodA was the only SOD identified in the extracellular fraction in B. subtilis.45,46 In addition, we purified the protein into homogeneity from the culture supernatant of B. amyloliquefaciens GF423. The specific activity of the purified SodA was 2360 U/mg. SodA activity was maintained in a pH range of 4.0 to 8.0. The enzyme was stable up to 40°C, and around 60% of activity was retained at 50°C. To improve expression of the sodA gene, B. amyloliquefaciens GF423 was mutagenised by UV irradiation. From the UV mutant library, we selected B. amyloliquefaciens GF424, a strain with 4.5-fold higher SOD activity than that of the wild type.
3.2. SOD activity of enteric-coated SOD from B. amyloliquefaciens
The enzyme activity of SOD partially purified by ultrafiltration was measured using a commercial kit, and the SOD activity value was 20.24 U/mg [non-coated SOD]. Enteric-coated SOD activity was 3.64 U/mg.
3.3. Antioxidant effect of BASOD in mice
To examine the antioxidative effect of BASOD, shellac-coated BASOD was administered to the irradiated mice, and the degree of change of antioxidant enzymes in the blood was measured. The levels of antioxidant enzymes, superoxide dismutase [SOD], catalase [CAT], and glutathione peroxidase [GPx], were higher in the blood of the BA SOD group than in the control. SOD, CAT, and GPx were also higher in the BA WT spore, BA HPSOD spore, BA WT spore + BA SOD, and BA HPSOD spore + BA SOD groups; levels in the BA ΔSODA spore group were not affected. Contrastingly, antioxidant enzymes of the group irradiated with γ-rays were decreased. In the BA SOD + irradiation group, antioxidant enzymes in the blood recovered to a level similar to that of the control group [Table 1]. These data show that BASOD has an antioxidative effect and can ameliorate oxidative stress.
Measurement of antioxidant enzymes in the irradiated group of mice.
| Activity | ||||||
|---|---|---|---|---|---|---|
| SOD [unit/mL] | Catalase [nmol/min/mL] | GPx [nmol/min/mL] | ||||
| 0 | 28 | 0 | 28 | 0 | 28 | |
| Control | 16.8 ± 0.03 | 19.7 ± 0.03 | 274.06 ± 0.1 | 243.3 ± 0.5 | 3170.4 ± 0.01 | 3146.9 ± 0.07 |
| BASOD | 15.5 ± 0.05 | 24.9 ± 0.07* | 382.3 ± 0.09 | 555.7 ± 0.1 | 2329.6 ± 0.02 | 2738.2 ± 0.05* |
| γ-irradiation [control] | 14.8 ± 0.1 | 10.8 ± 0.2 | 283.5 ± 0.02 | 141.1 ± 0.03 | 2207.8 ± 0.01 | 117.9 ± 0.05* |
| γ-irradiation + BASOD | 14.0 ± 0.07 | 18.2 ± 0.2 | 357.9 ± 0.01 | 316.3 ± 0.04* | 1964.0 ± 0.01 | 150.8 ± 0.05 |
| BA WT spore | 14.2 ± 0.33 | 19.9 ± 0.2 | 300.3 ± 0.8 | 447.5 ± 0.69 | 3217.8 ± 0.34 | 3809.9 ± 0.37 |
| BA ΔSODA spore | 14.8 ± 0.13 | 14.7 ± 0.12 | 300. ±0.24 | 305.2 ± 0.28 | 2821.8 ± 0.11 | 2839.2 ± 0.05 |
| BA HP SOD spore | 14.6 ± 0.14 | 22.5 ± 0.11* | 307.2 ± 0.62 | 550.1 ± 0.14* | 2844.1 ± 0.45 | 3578.4 ± 0.29 |
| BA WT spore +BASOD | 14.5 ± 0.05 | 26.1 ± 0.14* | 291.2 ± 0.89 | 557.8 ± 0.78* | 3103.1 ± 0.28 | 4132.2 ± 0.13* |
| BA HP SOD spore + BASOD | 14.8 ± 0.08 | 26.5 ± 0.02* | 320.4 ± 0.72 | 580.2 ± 0.99* | 3905.2 ± 0.22 | 5033.4 ± 0.2* |
| Activity | ||||||
|---|---|---|---|---|---|---|
| SOD [unit/mL] | Catalase [nmol/min/mL] | GPx [nmol/min/mL] | ||||
| 0 | 28 | 0 | 28 | 0 | 28 | |
| Control | 16.8 ± 0.03 | 19.7 ± 0.03 | 274.06 ± 0.1 | 243.3 ± 0.5 | 3170.4 ± 0.01 | 3146.9 ± 0.07 |
| BASOD | 15.5 ± 0.05 | 24.9 ± 0.07* | 382.3 ± 0.09 | 555.7 ± 0.1 | 2329.6 ± 0.02 | 2738.2 ± 0.05* |
| γ-irradiation [control] | 14.8 ± 0.1 | 10.8 ± 0.2 | 283.5 ± 0.02 | 141.1 ± 0.03 | 2207.8 ± 0.01 | 117.9 ± 0.05* |
| γ-irradiation + BASOD | 14.0 ± 0.07 | 18.2 ± 0.2 | 357.9 ± 0.01 | 316.3 ± 0.04* | 1964.0 ± 0.01 | 150.8 ± 0.05 |
| BA WT spore | 14.2 ± 0.33 | 19.9 ± 0.2 | 300.3 ± 0.8 | 447.5 ± 0.69 | 3217.8 ± 0.34 | 3809.9 ± 0.37 |
| BA ΔSODA spore | 14.8 ± 0.13 | 14.7 ± 0.12 | 300. ±0.24 | 305.2 ± 0.28 | 2821.8 ± 0.11 | 2839.2 ± 0.05 |
| BA HP SOD spore | 14.6 ± 0.14 | 22.5 ± 0.11* | 307.2 ± 0.62 | 550.1 ± 0.14* | 2844.1 ± 0.45 | 3578.4 ± 0.29 |
| BA WT spore +BASOD | 14.5 ± 0.05 | 26.1 ± 0.14* | 291.2 ± 0.89 | 557.8 ± 0.78* | 3103.1 ± 0.28 | 4132.2 ± 0.13* |
| BA HP SOD spore + BASOD | 14.8 ± 0.08 | 26.5 ± 0.02* | 320.4 ± 0.72 | 580.2 ± 0.99* | 3905.2 ± 0.22 | 5033.4 ± 0.2* |
Two-point check on the first and last days. Values are mean ± SD [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. *Significant differences [p <0.05] compared with Day 0 of each group.
BA WT spore, wild-type B. amyloliquefaciens GF423; BA ΔSOD A spore, SOD knock-out mutant B. amyloliquefaciens; BA HP SOD spore, high-SOD-producing strain B. amyloliquefaciens GF424; SOD, superoxide dismutase.
Measurement of antioxidant enzymes in the irradiated group of mice.
| Activity | ||||||
|---|---|---|---|---|---|---|
| SOD [unit/mL] | Catalase [nmol/min/mL] | GPx [nmol/min/mL] | ||||
| 0 | 28 | 0 | 28 | 0 | 28 | |
| Control | 16.8 ± 0.03 | 19.7 ± 0.03 | 274.06 ± 0.1 | 243.3 ± 0.5 | 3170.4 ± 0.01 | 3146.9 ± 0.07 |
| BASOD | 15.5 ± 0.05 | 24.9 ± 0.07* | 382.3 ± 0.09 | 555.7 ± 0.1 | 2329.6 ± 0.02 | 2738.2 ± 0.05* |
| γ-irradiation [control] | 14.8 ± 0.1 | 10.8 ± 0.2 | 283.5 ± 0.02 | 141.1 ± 0.03 | 2207.8 ± 0.01 | 117.9 ± 0.05* |
| γ-irradiation + BASOD | 14.0 ± 0.07 | 18.2 ± 0.2 | 357.9 ± 0.01 | 316.3 ± 0.04* | 1964.0 ± 0.01 | 150.8 ± 0.05 |
| BA WT spore | 14.2 ± 0.33 | 19.9 ± 0.2 | 300.3 ± 0.8 | 447.5 ± 0.69 | 3217.8 ± 0.34 | 3809.9 ± 0.37 |
| BA ΔSODA spore | 14.8 ± 0.13 | 14.7 ± 0.12 | 300. ±0.24 | 305.2 ± 0.28 | 2821.8 ± 0.11 | 2839.2 ± 0.05 |
| BA HP SOD spore | 14.6 ± 0.14 | 22.5 ± 0.11* | 307.2 ± 0.62 | 550.1 ± 0.14* | 2844.1 ± 0.45 | 3578.4 ± 0.29 |
| BA WT spore +BASOD | 14.5 ± 0.05 | 26.1 ± 0.14* | 291.2 ± 0.89 | 557.8 ± 0.78* | 3103.1 ± 0.28 | 4132.2 ± 0.13* |
| BA HP SOD spore + BASOD | 14.8 ± 0.08 | 26.5 ± 0.02* | 320.4 ± 0.72 | 580.2 ± 0.99* | 3905.2 ± 0.22 | 5033.4 ± 0.2* |
| Activity | ||||||
|---|---|---|---|---|---|---|
| SOD [unit/mL] | Catalase [nmol/min/mL] | GPx [nmol/min/mL] | ||||
| 0 | 28 | 0 | 28 | 0 | 28 | |
| Control | 16.8 ± 0.03 | 19.7 ± 0.03 | 274.06 ± 0.1 | 243.3 ± 0.5 | 3170.4 ± 0.01 | 3146.9 ± 0.07 |
| BASOD | 15.5 ± 0.05 | 24.9 ± 0.07* | 382.3 ± 0.09 | 555.7 ± 0.1 | 2329.6 ± 0.02 | 2738.2 ± 0.05* |
| γ-irradiation [control] | 14.8 ± 0.1 | 10.8 ± 0.2 | 283.5 ± 0.02 | 141.1 ± 0.03 | 2207.8 ± 0.01 | 117.9 ± 0.05* |
| γ-irradiation + BASOD | 14.0 ± 0.07 | 18.2 ± 0.2 | 357.9 ± 0.01 | 316.3 ± 0.04* | 1964.0 ± 0.01 | 150.8 ± 0.05 |
| BA WT spore | 14.2 ± 0.33 | 19.9 ± 0.2 | 300.3 ± 0.8 | 447.5 ± 0.69 | 3217.8 ± 0.34 | 3809.9 ± 0.37 |
| BA ΔSODA spore | 14.8 ± 0.13 | 14.7 ± 0.12 | 300. ±0.24 | 305.2 ± 0.28 | 2821.8 ± 0.11 | 2839.2 ± 0.05 |
| BA HP SOD spore | 14.6 ± 0.14 | 22.5 ± 0.11* | 307.2 ± 0.62 | 550.1 ± 0.14* | 2844.1 ± 0.45 | 3578.4 ± 0.29 |
| BA WT spore +BASOD | 14.5 ± 0.05 | 26.1 ± 0.14* | 291.2 ± 0.89 | 557.8 ± 0.78* | 3103.1 ± 0.28 | 4132.2 ± 0.13* |
| BA HP SOD spore + BASOD | 14.8 ± 0.08 | 26.5 ± 0.02* | 320.4 ± 0.72 | 580.2 ± 0.99* | 3905.2 ± 0.22 | 5033.4 ± 0.2* |
Two-point check on the first and last days. Values are mean ± SD [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. *Significant differences [p <0.05] compared with Day 0 of each group.
BA WT spore, wild-type B. amyloliquefaciens GF423; BA ΔSOD A spore, SOD knock-out mutant B. amyloliquefaciens; BA HP SOD spore, high-SOD-producing strain B. amyloliquefaciens GF424; SOD, superoxide dismutase.
3.4. Effect of BA SOD on DSS-induced IBD in mice
In mouse-modelled DSS-induced ulcerative colitis, BASOD, BA WT spores, and BA HP SOD spores reduced mortality, and BA ΔSODA spores had no effect.
Figure 2 shows the outcomes in DSS-treated mice. Survival was the highest in the group treated with both wild-type and high-producing spores [>85%]; survival was greater than 65%, 55%, and 40% of mice treated with only oral SOD, high-producing spore, or wild-type spore, respectively. Mice fed only DSS or DSS + BA ΔSODA spore survived at a rate of 30% [Figure 2A, B]. Weight loss also differed; the loss rates of the group fed DSS only and DSS + BA ΔSODA spore were greater than those of the control group. Other groups also had greater weight loss than the control group, but loss was less severe [Figure 2C, D].
Protective effect of superoxide dismutase from B. amyloliquefaciens [BA SOD] and BA spores on [A and B] survival rate and [C and D] weight change of the dextran sulphate sodium [DSS]-treated mice. Data are presented as mean ± standard deviation [SD] [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. Significant differences [*p <0.05, **p <0.01, ***p <0.005] are compared with the control group. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant of B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
Protective effect of superoxide dismutase from B. amyloliquefaciens [BA SOD] and BA spores on [A and B] survival rate and [C and D] weight change of the dextran sulphate sodium [DSS]-treated mice. Data are presented as mean ± standard deviation [SD] [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. Significant differences [*p <0.05, **p <0.01, ***p <0.005] are compared with the control group. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant of B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
BA SOD and SOD-producing BA spore markedly improved clinical signs of DSS-induced colitis, including rectal bleeding [Figure 3A, B] and diarrhoea [Figure 3C, D]. The colon length of the group fed only DSS was shorter than that of groups fed DSS + BASOD, DSS + BA spore, and DSS + BASOD + BA spore [Figure 3E]. These data show that BASOD and SOD-producing BA spores ameliorate the development of DSS-induced colitis.
Protective effect of superoxide dismutase from B. amyloliquefaciens [BASOD] and BA spores on [A and B] diarrhea, [C and D] rectal bleeding, and [E] colon length of the dextran sulphate sodium [DSS]-induced mice. Data are presented as means ± standard deviation [SD]: [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. Significant differences [*p <0.05, **p <0.01, ***p <0.005] of [A], [B], [C], and [D] are compared twith the control group, and that of [E] is compared with the DSS group. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
Protective effect of superoxide dismutase from B. amyloliquefaciens [BASOD] and BA spores on [A and B] diarrhea, [C and D] rectal bleeding, and [E] colon length of the dextran sulphate sodium [DSS]-induced mice. Data are presented as means ± standard deviation [SD]: [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. Significant differences [*p <0.05, **p <0.01, ***p <0.005] of [A], [B], [C], and [D] are compared twith the control group, and that of [E] is compared with the DSS group. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
3.5. Histology of colon tissue
The control group showed histologically normal colonic mucosa. In mice treated with DSS only, the colonic mucosa was severely damaged and a highly inflamed mucosa was observed. A damaged colonic mucosa was also observed in mice treated with DSS and BA ΔSODA spore. In mice treated with DSS and BA SOD, BA WT spore, and/or BA HPSOD spore, some damaged colonic mucosa was observed, but the level was much lower than that of mice treated with DSS alone [Figure 4A]. In the mice treated with DSS, inflammation was observed in mucosal and submucosal layers, and the mucous membrane adjacent to the site of ulceration had increased oedema with extensive cell depletion, as compared with the control mice [Figure 4B]. After the dietary treatment of BASOD, BA WT spore, and/or BA HPSOD spore, histological analysis revealed a reduction of morphological signs of cell damage. These data indicate that BA SOD and BA spores producing SOD reduce mucosal damage in DSS-induced colitis.
Effect of B. amyloliquefaciens superoxide dismutase [BA SOD] and BA spores on mucosal damage in colitis mice. [A] Representative photographs of haematoxylin and eosin-stained colonic tissues from control, dextran sulphate sodium [DSS], DSS + BA WT spore, DSS + BA ΔSODA spore, DSS + BA HPSOD spore, DSS + BA WT spore + BA SOD, DSS + BA HPSOD pore + BA SOD, and DSS + BA SOD groups. [B] Change in histological score according to the criteria defined in the ‘Materials and Methods.’ Data are presented as mean ± standard deviation [SD] [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. Significant differences [*p <0.05, #p <0.01] are compared with the DSS group.
Effect of B. amyloliquefaciens superoxide dismutase [BA SOD] and BA spores on mucosal damage in colitis mice. [A] Representative photographs of haematoxylin and eosin-stained colonic tissues from control, dextran sulphate sodium [DSS], DSS + BA WT spore, DSS + BA ΔSODA spore, DSS + BA HPSOD spore, DSS + BA WT spore + BA SOD, DSS + BA HPSOD pore + BA SOD, and DSS + BA SOD groups. [B] Change in histological score according to the criteria defined in the ‘Materials and Methods.’ Data are presented as mean ± standard deviation [SD] [n = 7 for each group]. Statistical analysis was performed using two-tailed, unpaired Student’s t-test. Significant differences [*p <0.05, #p <0.01] are compared with the DSS group.
3.6. Effect of BA SOD on antioxidant enzymes in serum samples of DSS-induced colitis
The serum SOD level of mice fed only DSS was decreased compared with that of the control group, and the SOD level of mice fed DSS with BA spore or BASOD was higher than that in mice fed only DSS [Figure 5A]. CAT and GPx levels of mice fed only DSS were lower than those in the control group, whereas the levels in mice fed DSS with BA spores or BASOD were higher than those in DSS-only mice [Figure 5B, C]. These data show that BASOD most likely helps to improve colitis by affecting the body’s antioxidant enzymes.
Effect of B. amyloliquefaciens superoxide dismutase [BA SOD] and BA spores on the antioxidant enzymes in mice with dextran sulphate sodium [DSS]-induced colitis. [A] SOD activity in serum of mice. [B] Catalase [CAT] activity in serum of mice. [C] Glutathione peroxidase [GPx] activity in serum of mice. Statistical analysis was performed using two-tailed, unpaired Student’s t-test: significant differences [*p <0.05, #p <0.01] compared with the DSS group. Values are mean ± standard deviation [SD] [n = 7 for each group]. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
Effect of B. amyloliquefaciens superoxide dismutase [BA SOD] and BA spores on the antioxidant enzymes in mice with dextran sulphate sodium [DSS]-induced colitis. [A] SOD activity in serum of mice. [B] Catalase [CAT] activity in serum of mice. [C] Glutathione peroxidase [GPx] activity in serum of mice. Statistical analysis was performed using two-tailed, unpaired Student’s t-test: significant differences [*p <0.05, #p <0.01] compared with the DSS group. Values are mean ± standard deviation [SD] [n = 7 for each group]. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
3.7. Effect of BA SOD on inflammatory cytokines in DSS-induced colitis
Levels of pro-inflammatory cytokines TNFα and IL1β were higher in mice fed only DSS and lower in mice fed DSS with BA SOD or BA spores [Figure 6A, B]. Levels of IL6, an anti-inflammatory cytokine, decreased in mice fed only DSS and increased in mice fed DSS with BA SOD or BA spores [Figure 6C]. These data indicate that BASOD modulates inflammatory cytokines and may thereby improve IBD symptoms.
Effect of B. amyloliquefaciens superoxide dismutase [BA SOD] and BA spores on inflammatory cytokines in mice with dextran sulphate sodium [DSS]-induced colitis. [A] Pro-inflammatory cytokine tumour necrosis factor [TNF]-α in serum of mice. [B] Pro-inflammatory cytokine IL-1β in serum of mice. [C] Anti-inflammatory cytokine IL6 in serum of mice. Statistical analysis was performed using two-tailed, unpaired Student’s t-test: significant differences [*p <0.05, #p <0.01] compared with the DSS group. Values are mean ± standard deviation [SD] [n = 7 for each group]. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
Effect of B. amyloliquefaciens superoxide dismutase [BA SOD] and BA spores on inflammatory cytokines in mice with dextran sulphate sodium [DSS]-induced colitis. [A] Pro-inflammatory cytokine tumour necrosis factor [TNF]-α in serum of mice. [B] Pro-inflammatory cytokine IL-1β in serum of mice. [C] Anti-inflammatory cytokine IL6 in serum of mice. Statistical analysis was performed using two-tailed, unpaired Student’s t-test: significant differences [*p <0.05, #p <0.01] compared with the DSS group. Values are mean ± standard deviation [SD] [n = 7 for each group]. DSS, only DSS; BA WT spore, B. amyloliquefaciens GF423; BA ΔSODA spore, SOD knock-out mutant B. amyloliquefaciens; BA HPSOD spore, B. amyloliquefaciens GF424.
4. Discussion
ROS have been implicated in a range of pathologies, such as cancer, cardiovascular diseases [CVDs], degenerative diseases, and infectious diseases. For many scientists studying ROS-related disorders, the manipulation of antioxidant levels offers the possibility to ameliorate particular conditions.3 Most antioxidants externally provided from dietary sources are quickly saturated and exhausted, whereas SOD keeps eliminating ROS. Therefore, the therapeutic value of oral administration of a combination of melon SOD and a vegetable polymer [gliadin] has been evaluated in many studies.3 To further clarify the importance of dietary SOD–gliadin administration, additional large-scale clinical trials are proceeding. The use of bacillus SODs could make supplementation with SODs more economical and effective.
Bacillus SODs have been studied from a biochemical and physiological perspective. Mn-SOD [encoded by sodA gene] is essential for resistance to oxidative stress in growing and sporulating cells of B. subtilis.5 Although many beneficial effects related to increasing SOD levels in animals and humans have been found after administration of bacillus cells, no research has been conducted using bacillus SOD itself, which might be due to its low production level.
Although Mn-SOD is classified as an intracellular protein, it is also consistently found in extracellular protein samples of many bacilli.5B. amyloliquefaciens has three sod genes: sodA, sodC, and sodF. The negligible activity of the sodA knock-out mutant demonstrates that that the SOD activity of B. amyloliquefaciens was derived from secreted Mn-SOD encoded by the sodA gene. We therefore isolated a high-SOD-producing strain using UV mutagenesis and developed the downstream process of SOD production. Gliadin has been shown to interact with intestinal epithelial cells and cross the intestinal epithelial barrier.47,48 Thus, we chose shellac as an enteric coating material to prolong the half-life of SOD. The specific activity of partially purified SOD using ultrafiltration was 20.24 U/mg before enteric coating with shellac and 3.64 U/mg after coating; these specific activities are sufficient for commercial use. The reduced mortality and morbidity of mice treated with purified SOD and SOD spores, especially combinations thereof, demonstrate the clinical benefits of this level of SOD activity.
Our results indicated that SOD supplementation not only promoted antioxidant defences [increased SOD, catalase, and GPx activities] but also improved cell resistance to the oxidative stress of γ-irradiation. This result indicates that B. amyloliquefaciens SOD could be an alternative ingredient for dietary supplementation for anti-ageing and well-being in addition to ulcerative colitis. However, the mechanism by which exogenous bacillus SOD promotes antioxidant defences in the body will need to be investigated in further studies.
The scientific community has sought novel therapeutic alternatives to fight IBD and mucositis. As dysbiosis plays a key role in the pathogenesis of both diseases, the modulation of the patient microbiota via the administration of probiotic bacteria has been proposed. Experiments with lactic acid bacteria [LAB] strains engineered to express anti-inflammatory proteins have shown promise. Mouse transforming growth factor β, human interleukin-10, human trefoil factor I, human elafin, mouse cathelicidin, mouse leukocyte protease inhibitor, and human 15-lipoxygenase-1 including SODs from Lactococcus lactis and B. subtilis, have been produced in different strains of lactic acid bacteria and tested in several inflammatory conditions.49
The ability of SOD to control the ROS level within cells renders it a highly effective therapeutic protein for the treatment of IBD.50 Intestinal inflammation is accompanied by massive ROS production by activated macrophages and neutrophils, which contributes to inflammation-associated tissue destruction. Many studies have documented that exogenous SOD can attenuate inflammation by exerting antioxidative effects on a variety of experimental models,33 and using GRAS strains as vehicles to deliver bioactive molecules to the gastrointestinal tract has been considered a promising approach for the treatment of many diseases, including IBD.32 Although bacillus strains have been considered soil organisms for which endospore formation provides a means to ensure long-term survival in the environment, bacillus spores are not transient passengers of the gastrointestinal tract but have adapted to carry out their entire life cycle within this environment.51 Therefore, the bacillus cytoplasmic protein, which includes SOD, could be released into the gastrointestinal tract through the sporulation life cycle. Moreover, as mentioned above, B. amyloliquefaciens SOD is found extracellularly in liquid culture. Even if the role of SOD in B. polyfermenticus has not been elucidated, the probiotic effect of B. polyfermenticus in intestinal inflammation by suppressing apoptosis and promoting epithelial cell proliferation and migration has been reported.52 Our results are consistent with these reports. As expected, wild-type B. amyloliquefaciens had a beneficial effect in DSS-induced mouse model, and the effect of the SOD high-producer strain [GF424] was proportional to its SOD productivity. Moreover, co-administration of SOD protein with B. amyloliquefaciens spore showed a synergistic therapeutic effect.
Our results are an important step in increasing the accessibility of SOD to IBD patients. Microbial suspension culture is a more convenient mode of mass cultivation of cells, large-scale production, and recovery of enzyme, than plant cultivation. The production of bacillus SOD could be more economical than melon-derived SOD. Moreover, the entire production could take place under highly controlled conditions.
Our findings suggest that the co-administration of enteric-coated SOD with the wild-type strain or the high-SOD-producing strain spores of B. amyloliquefaciens is a promising candidate to alleviate oxidative stress and inflammation response in humans, without the issues regarding the use of genetically modified bacteria as a delivery vehicle of therapeutic protein. However, the acute DSS colitis model is generally considered as a model for acute intestinal inflammation rather than an IBD model. The chronic DSS colitis or IL-10 knock-out model would need to be tested.
Funding
No funding was provided for this work.
Conflict of Interest
The authors declare that they have no competing interests.
Author Contributions
JEK: acquisition of data; analysis and interpretation of data; drafting of the manuscript; statistical analysis. HDK: acquisition of data. SYP: acquisition of data; analysis and interpretation of data. JGP: study concept; critical revision of the manuscript for important intellectual content. JHK: analysis and interpretation of data; critical revision of the manuscript for important intellectual content. DYY: study concept and design; analysis and interpretation of data; critical revision of the manuscript for important intellectual content; study supervision. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Acknowledgments
The authors would like to thank Dr Hee-Gu Lee of the Korea Research Institute of Bioscience and Biotechnology [KRIBB] for his excellent technical assistance during this study.
References
J
, JM
, JP
, R
. Superoxide dismutase administration, a potential therapy against oxidative stress related diseases: several routes of supplementation and proposal of an original mechanism of action
. 2013
;:2718
–.T
, M
. Superoxide dismutases: role in redox signaling, vascular function, and diseases
. 2011
;: 1583
–.S
. Therapeutic value of oral supplementation with melon superoxide dismutase and wheat gliadin combination
. 2015
;: 430
–.IA
, DE
. Superoxide dismutases - a review of the metal-associated mechanistic variations
. 2010
;:263
–.P
, HE
, YJ
, CD
, PC
, IT
. Structure of Bacillus subtilis superoxide dismutase
. 2007
;:1003
–.M
, J
, CC
. Effect of heavy metals on growth response and antioxidant defense protection in Bacillus cereus
. 2014
;:1201
–.S
, PK
, TK
. Influence of heavy metals on the activity of antioxidant enzymes in the metal resistant strains of Ochrobactrum and Bacillus sp
. 2013
;:1033
–.M
, SS
, HR
, S
, J
, MH
. Erratum to: effect of probiotic Bacillus coagulans and Lactobacillus plantarum on alleviation of mercury toxicity in rat
. 2017
;:214
.X
, E
, L
et al. Repair effects of exogenous SOD on Bacillus subtilis against gamma radiation exposure
. 2013
;:259
–.H
, S
, Y
et al. Dietary administration of Bacillus subtilis HAINUP40 enhances growth, digestive enzyme activities, innate immune responses and disease resistance of tilapia, Oreochromis niloticus
. 2017
;:326
–.S
, K
, Y
et al. Comparative evaluation of dietary probiotics Bacillus subtilis WB60 and Lactobacillus plantarum KCTC3928 on the growth performance, immunological parameters, gut morphology and disease resistance in Japanese eel, Anguilla japonica
. 2017
;:201
–.CN
, XF
, WN
et al. Combined effects of dietary fructooligosaccharide and Bacillus licheniformis on innate immunity, antioxidant capability and disease resistance of triangular bream [Megalobrama terminalis]
. 2013
;:1380
–.CH
, CH
, SW
, W
. Dietary administration of the probiotic, Bacillus subtilis E20, enhances the growth, innate immune responses, and disease resistance of the grouper, Epinephelus coioides
. 2012
;:699
–.K
, Q
, J
, J
, L
, T
. Supplemental effects of probiotic Bacillus subtilis fmbJ on growth performance, antioxidant capacity, and meat quality of broiler chickens
. 2017
;:74
–.Y
, Y
, J
, M
, Q
, Y
. Biocontrol agent Bacillus amyloliquefaciens LJ02 induces systemic resistance against cucurbits powdery mildew
. 2015
;:883
.. Carbohydrase and Protease Enzyme Preparations Derived From Bacillus Subtilis or Bacillus Amyloliquefaciens; Affirmation of Gras Status as Direct Food Ingredients
. 1999. https://federalregister.Gov/a/99-10011 Accessed .. Opinion of the scientific committee on a request from EFSA related to a generic approach to the safety assessment by EFSA of microorganisms used in food/feed and the production of food/feed additives
. 2005
;:1
–.HT
, NN
, OM
, et al. Effects of the dietary supplementation of mixed probiotic spores of Bacillus amyloliquefaciens 54a, and Bacillus pumilus 47b on growth, innate immunity and stress responses of striped catfish [Pangasianodon hypophthalmus]
. 2017
;:391
–.ST
, D
, B
, B
, J
. Paraprobiotic preparation from Bacillus amyloliquefaciens FPTB16 modulates immune response and immune relevant gene expression in Catla catla [Hamilton, 1822]
. 2017
;:35
–.H
, J
, Y
et al. Two novel exopolysaccharides from Bacillus amyloliquefaciens c-1: Antioxidation and effect on oxidative stress
. 2015
;:298
–.Y
, H
, YP
et al. Bacillus amyloliquefaciens supplementation alleviates immunological stress in lipopolysaccharide-challenged broilers at early age
. 2015
;:1504
–.X
, X
, Y
, H
, A
, H
. Effect of Bacillus amyloliquefaciens-based direct-fed microbial on performance, nutrient utilization, intestinal morphology and cecal microflora in broiler chickens
. 2015
;:239
–.ST
, M
, HS
, HJ
, YJ
, CJ
. Effects of Bacillus amyloliquefaciens as a probiotic strain on growth performance, cecal microflora, and fecal noxious gas emissions of broiler chickens
. 2014
;:1963
–.G
, L
, JJ
, M
, M
. Effects of dietary supplementation of Bacillus amyloliquefaciens CECT 5940 and Enterococcus faecium CECT 4515 in adult healthy dogs
. 2013
;:406
–.A
, K
, S
, D
. Effects of potential probiotic Bacillus amyloliquefaciens [corrected] FPTB16 on systemic and cutaneous mucosal immune responses and disease resistance of catla [Catla catla]
. 2013
;:1547
–.VI
, N
, P
, S
. Isolation and characterization of putative probiotic bacterial strain, Bacillus amyloliquefaciens, from North East Himalayan soil based on in vitro and in vivo functional properties
. 2011
;:175
–.GY
, SR
. Future biologic targets for IBD: potentials and pitfalls
. 2010
;:110
–.Q
, H
, Y
et al. A superoxide dismutase/catalase mimetic nanomedicine for targeted therapy of inflammatory bowel disease
. 2016
;:206
–.B
, A
, RJ
. Genetics and pathogenesis of inflammatory bowel disease
. 2011
;:307
–.L
, SR
, KP
et al. Increased oxidative stress and decreased antioxidant defenses in mucosa of inflammatory bowel disease
. 1996
;:2078
–.E
, D
, H
, K
. Evaluation of serum trace element levels and superoxide dismutase activity in patients with inflammatory bowel disease: translating basic research into clinical application
. 2017
;:235
–.SA
, P
, K
et al. Host lysozyme-mediated lysis of Lactococcus lactis facilitates delivery of colitis-attenuating superoxide dismutase to inflamed colons
. 2015
;:7803
–.CL
, J
, XT
, H
, XF
, SY
. Superoxide dismutase recombinant Lactobacillus fermentum ameliorates intestinal oxidative stress through inhibiting NF-κB activation in a trinitrobenzene sulphonic acid-induced colitis mouse model
. 2014
;:1621
–.JG
, S
, A
et al. Use of superoxide dismutase and catalase producing lactic acid bacteria in TNBS induced Crohn’s disease in mice
. 2011
;:287
–.IM
, JM
, JM
, TR
, HM
, DS
. Anti-inflammatory properties of Lactobacillus gasseri expressing manganese superoxide dismutase using the interleukin 10-deficient mouse model of colitis
. 2007
;:G729
–.W
, A
, A
et al. Improvement of an experimental colitis in rats by lactic acid bacteria producing superoxide dismutase
. 2006
;:1044
–.W
, J
. Anti-inflammatory properties of lactic acid bacteria producing superoxide dismutase
. 2008
;:G353; author reply G354
.AM
, N
, P
. Plasmids for ectopic integration in Bacillus subtilis
. 1996
;:57
–.M
, R
. Easy cloning of mini-Tn10 insertions from the Bacillus subtilis chromosome
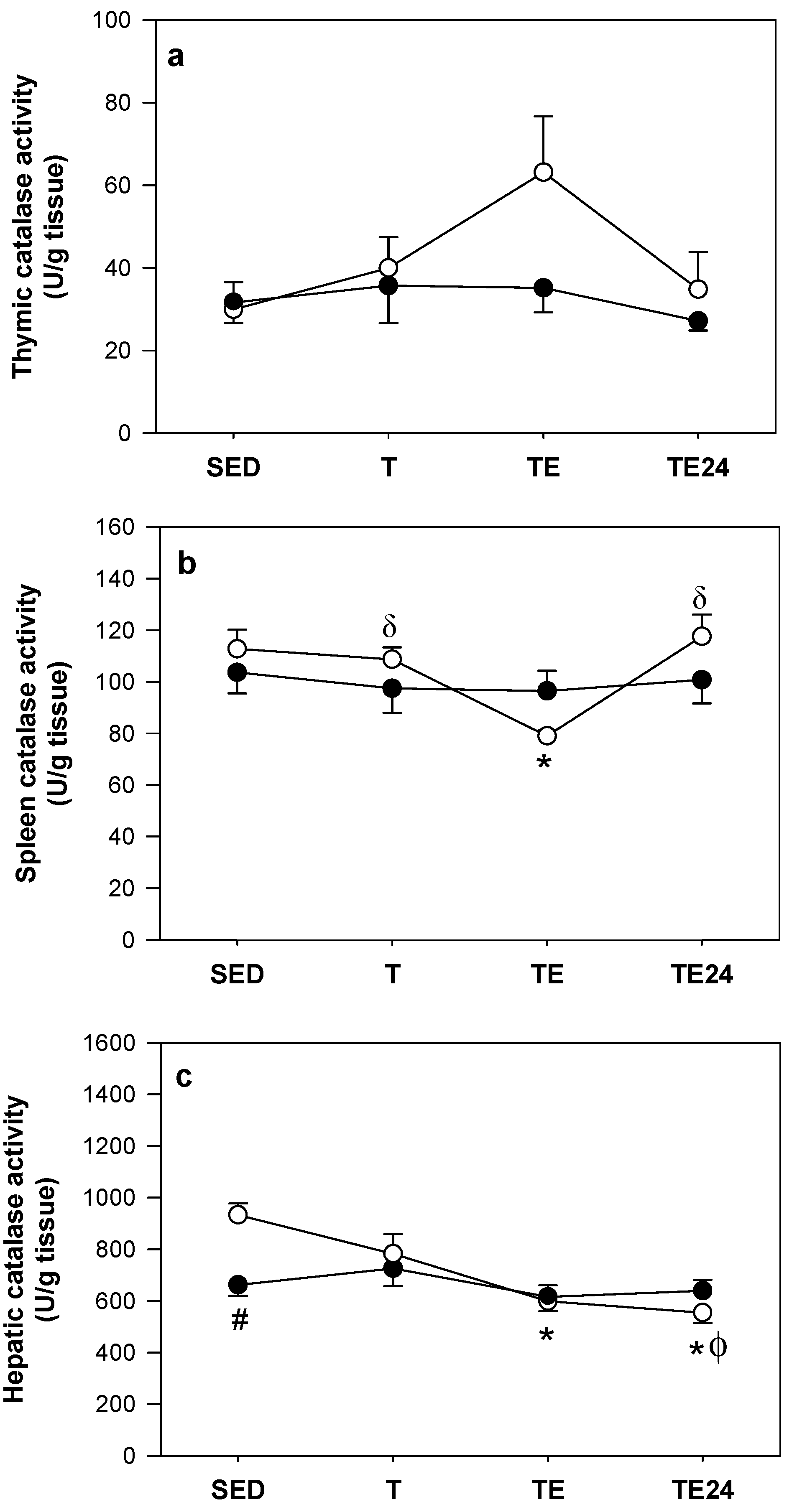
1994
;:1761
–.GQ
, P
, Y
, AH
, N
, TY
. Enhancing electro-transformation competency of recalcitrant Bacillus amyloliquefaciens by combining cell-wall weakening and cell-membrane fluidity disturbing
. 2011
;:130
–.VD
, KA
. . Mutat Res
;82
:–61
.MB
, RD
, WB
et al. Bacillus subtilis endospores at high purity and recovery yields: optimization of growth conditions and purification method
. 2013
;:279
–.R
, AW
. Role of TNF receptors, TNFR1 and TNFR2, in dextran sodium sulfate-induced colitis
. 2009
;:1515
–.RD
, TK
, PC
et al. InterPro in 2017-beyond protein family and domain annotations
. 2017
;:D190
–.TR
, H
, R
, EL
, M
, JM
. MscL of Bacillus subtilis prevents selective release of cytoplasmic proteins in a hypotonic environment
. 2009
;:1033
–.H
, JM
, S
, M
. Proteomic survey through secretome of Bacillus subtilis
. 2006
;:179
–.J
, EL
, B
, E
, A
, A
. Effect of gliadin on permeability of intestinal biopsy explants from celiac disease patients and patients with non-celiac gluten sensitivity
. 2015
;:1565
–.S
, R
, M
et al. Gliadin, zonulin and gut permeability: Effects on celiac and non-celiac intestinal mucosa and intestinal cell lines
. 2006
;:408
–.RDO
, FLR
, A
et al. Use of wild type or recombinant lactic acid bacteria as an alternative treatment for gastrointestinal inflammatory diseases: a focus on inflammatory bowel diseases and mucositis
. 2017
;:800
.A
, J
. Review article: the role of oxidative stress in pathogenesis and treatment of inflammatory bowel diseases
. 2014
;:605
–.NK
, NQ
, HA
et al. The intestinal life cycle of Bacillus subtilis and close relatives
. 2006
;:2692
–.E
, YJ
, C
, SH
. Bacillus polyfermenticus ameliorates colonic inflammation by promoting cytoprotective effects in colitic mice
. 2009
;:1848
–.Copyright © 2018 European Crohn’s and Colitis Organisation (ECCO). Published by Oxford University Press. All rights reserved. For permissions, please email: [email protected]
This article is published and distributed under the terms of the Oxford University Press, Standard Journals Publication Model (https://academic.oup.com/journals/pages/about_us/legal/notices)

